Questions
Question 1
A mixture of two miscible liquids K and M with close boiling points is separated using the apparatus shown in the figure below.
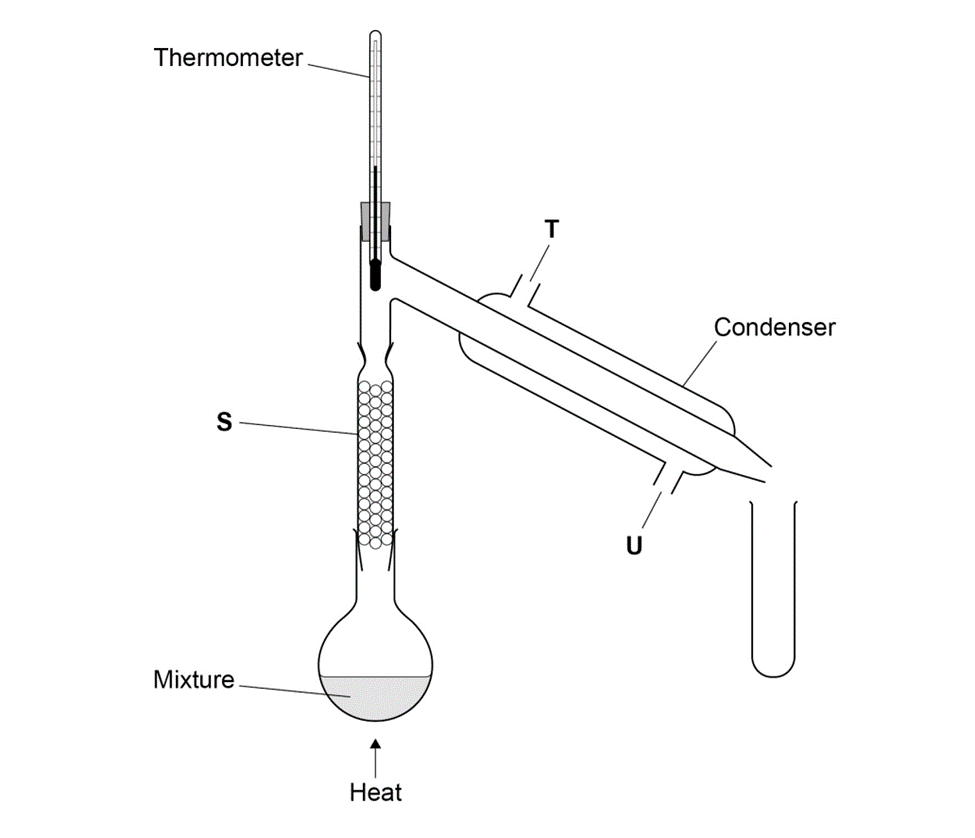
Label the parts indicated S, T and U.
State the purpose of the fractionating column packed with glass beads.
Explain why fractional distillation is preferred to simple distillation for separating K and M.
K has the lower boiling point. Predict which component (K or M) is collected first and justify your answer.
Explain why, for two isomers of similar molar mass, a more highly branched isomer tends to distill first. Refer to intermolecular forces/volatility.
After collecting a fraction believed to be an alcohol, identify one diagnostic IR absorption that would confirm the presence of an –OH group and one that would distinguish an aldehyde/ketone by their C=O group. Give typical wavenumber ranges.