Questions
Question 1
The figure below shows a branched alkene drawn as a skeletal formula.
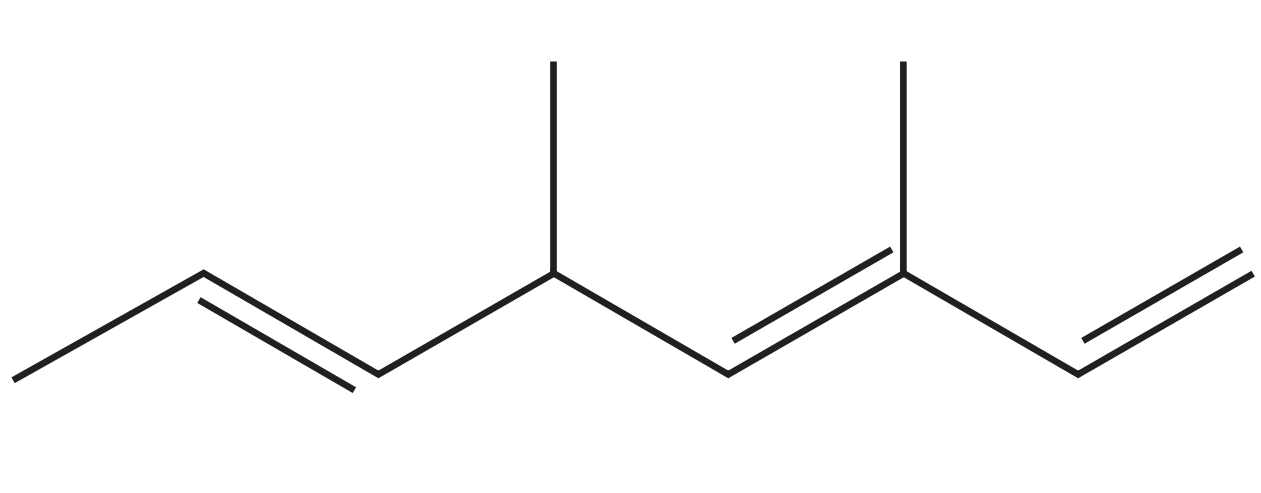
Determine the systematic IUPAC name of the molecule. Your name must identify the parent chain, the positions of all C=C bonds, and all substituents with correct locants.
State the total degree of unsaturation for this molecule.
When the alkene reacts rapidly with HBr in the dark and without peroxides, state the overall reaction type.
The same alkene reacts with HBr in the presence of peroxides and UV light. Predict the major constitutional product and justify your choice.
Deduce how many of the C=C bonds can exhibit E/Z stereoisomerism, and explain briefly.
Question 2
The graph below shows concentration–time data for the equilibrium between a cobalt(II) aqua complex and a chloro complex. At time t₁, aqueous AgNO₃ is added. Consider the equilibrium at 25 °C:
[Co(H₂O)₆]²⁺(aq) + 4 Cl⁻(aq) ⇌ [CoCl₄]²⁻(aq) + 6 H₂O(l)
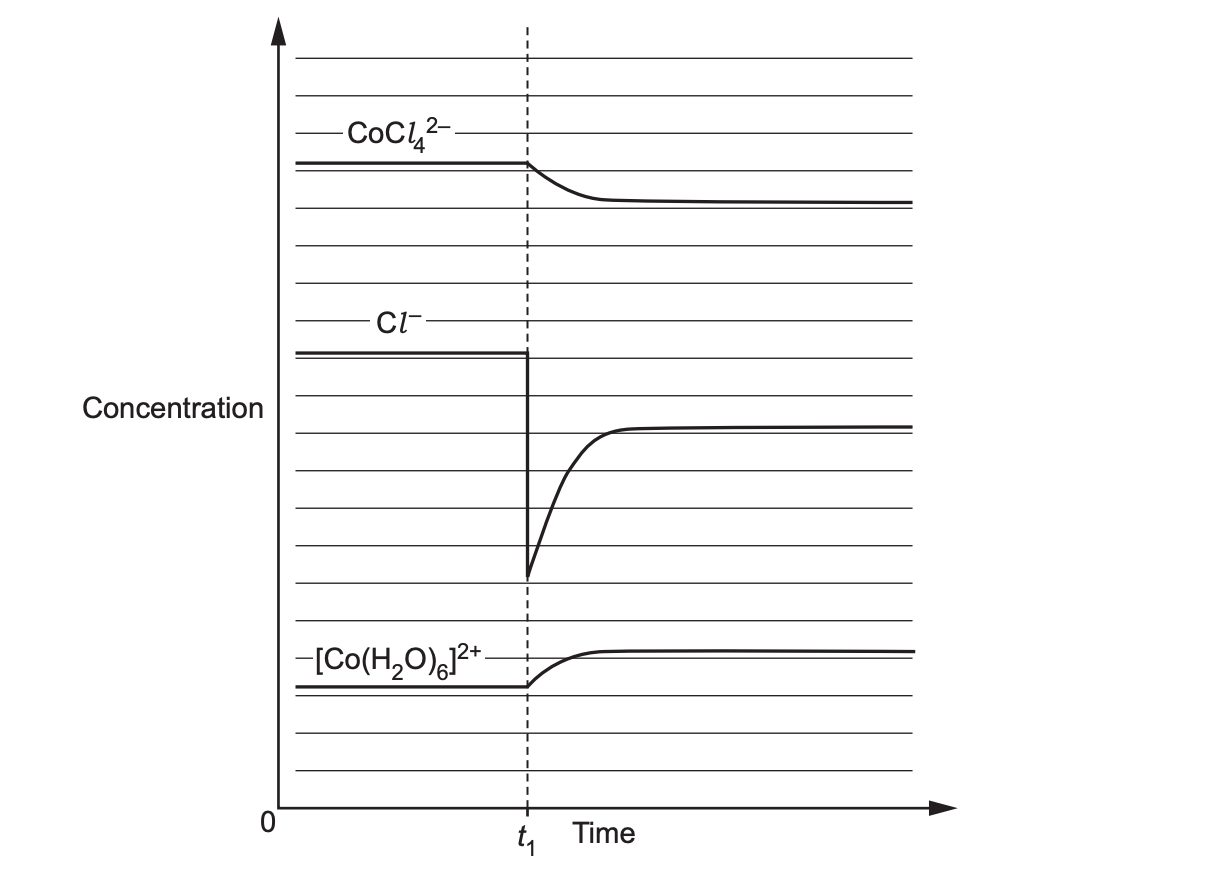
Using the graph, describe what happens to [Cl⁻], [[CoCl₄]²⁻] and [[Co(H₂O)₆]²⁺] immediately after t₁ and after a new equilibrium is established.
Write the equilibrium constant expression, Kc, for the reaction above.
Explain why adding AgNO₃ at t₁ shifts the equilibrium as shown in the graph. Refer to precipitation of AgCl and Le Châtelier’s principle.
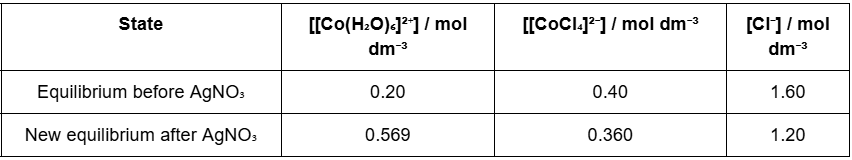
Use the data table below to determine Kc at 25 °C. Show working. Comment briefly on the two values you obtain.
The forward reaction is endothermic. If the mixture is warmed at constant volume, predict the change (increase, decrease or no change) in (i) the value of Kc and (ii) the proportion of [[CoCl₄]²⁻] at equilibrium.