Carbon chemistry and polymer reactions
Carbon forms covalent bonds and can make up to 4 bonds in total.
This allows huge diversity of biological molecules: straight chains, branched chains, single rings, and multiple rings.
Condensation reactions join monomers to make polymers, with water removed.
Hydrolysis reactions break polymers into monomers, with water added.
Must know examples of biological polymers: polysaccharides, polypeptides, nucleic acids.
Exam link: be able to identify whether a reaction shown is condensation or hydrolysis.
Monosaccharides
Monosaccharides are single sugar units.
Must recognize pentoses and hexoses from ring-form molecular diagrams.
Glucose is the key example for this topic.
Why glucose is useful biologically:
Soluble → easy to transport.
Transportable → can move in fluids.
Chemically stable → suitable for short-term use.
Releases a useful energy yield when oxidized in respiration.
Exam focus: identify monosaccharide structure from a diagram and link properties to function.
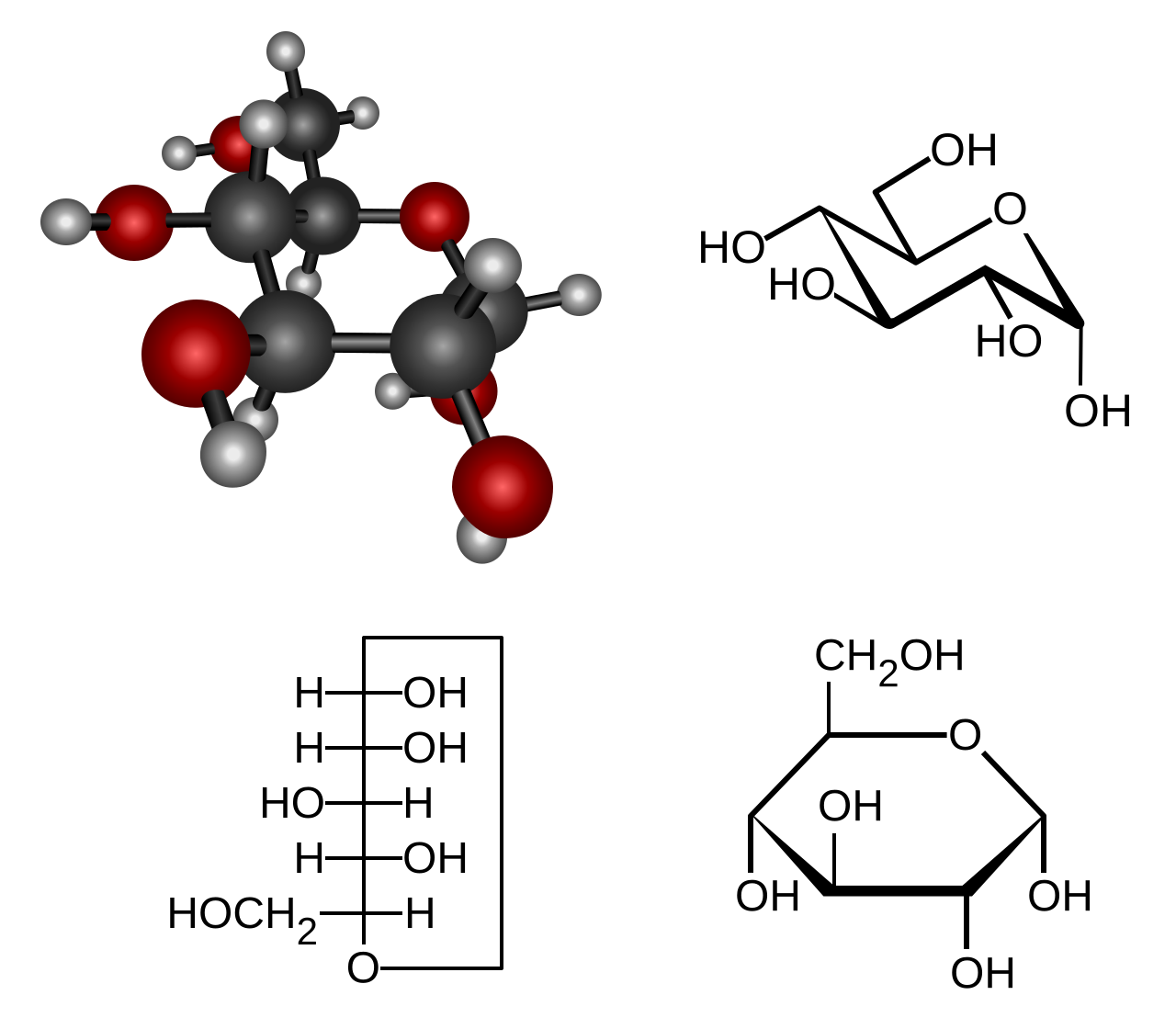
This diagram shows the ring structure of α-glucose, the monomer used to build starch and glycogen. It is useful for recognizing the hexose ring form expected in IB diagram questions. Source
Polysaccharides for energy storage
Polysaccharides are polymers of monosaccharides.
Starch is the main energy storage polysaccharide in plants.
Glycogen is the main energy storage polysaccharide in animals.
Both are suited to storage because they are:
Compact due to coiling and branching.
Relatively insoluble because of large molecular size.
Easy to build by condensation and break down by hydrolysis.
The monomers added/removed are α-glucose monomers.
Exam comparisons:
Starch = storage in plants.
Glycogen = storage in animals.
Both are better storage molecules than glucose because they are compact and less osmotically active due to their large size.
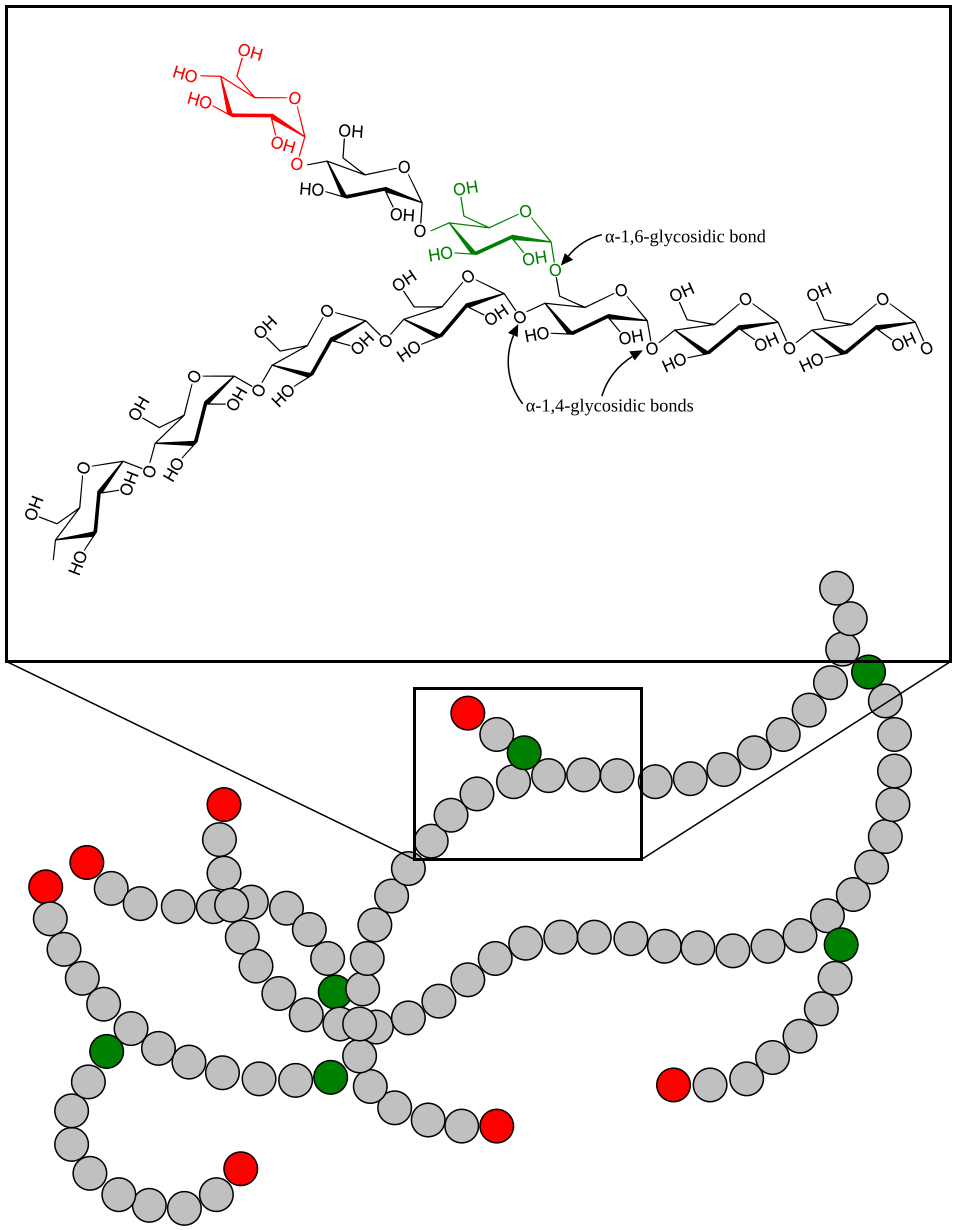
This diagram shows glycogen as a highly branched polymer of glucose. The branching helps explain why glycogen is compact and why glucose units can be added or removed rapidly during energy storage and mobilization. Source
Cellulose as a structural polysaccharide
Cellulose is a structural polysaccharide in plant cell walls.
Made from β-glucose, not α-glucose.
In cellulose, adjacent β-glucose monomers have alternating orientation.
This produces straight chains, not coiled chains.
Straight chains can be grouped into bundles.
Chains are cross-linked by hydrogen bonds.
This gives high tensile strength, making cellulose ideal for support in plants.
Key exam contrast:
Starch/glycogen → energy storage, coiled/branched, made from α-glucose.
Cellulose → structural support, straight chains, made from β-glucose.
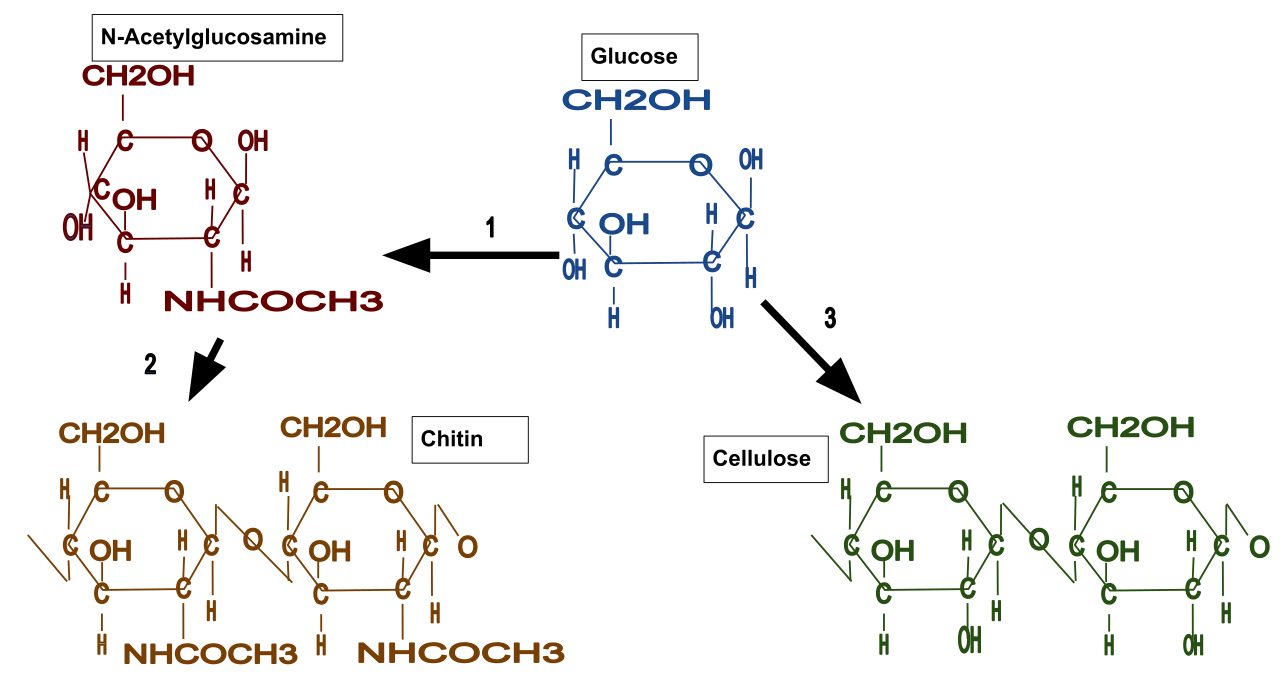
This illustration shows how glucose units form cellulose chains, which then assemble into microfibrils and larger fibres. It helps explain why cellulose has a straight, strong structure suitable for plant support. Source
Glycoproteins and cell recognition
Glycoproteins are proteins with carbohydrate chains attached.
They are important in cell–cell recognition.
A required example is ABO antigens on cell surfaces.
Exam point: different carbohydrate markers on cell surfaces allow cells to be recognized as different types.
Lipids: key properties
Lipids are biological substances that dissolve in non-polar solvents.
They are only sparingly soluble in water because they are largely hydrophobic.
Lipid groups named in the syllabus: fats, oils, waxes, steroids.
Core exam idea: hydrophobicity explains many lipid functions, especially storage and membrane formation.
Triglycerides and fatty acids
Triglycerides form by condensation reactions.
One glycerol joins to 3 fatty acids.
Triglycerides are used for long-term energy storage and thermal insulation.
Stored in adipose tissue.
Why triglycerides are good energy stores:
Energy-dense.
Hydrophobic and therefore insoluble, so they do not affect osmosis much.
Good for long-term storage.
Help with thermal insulation, especially important in animals living in cold habitats.
Saturated fatty acids: have no C=C double bonds.
Monounsaturated fatty acids: have 1 C=C double bond.
Polyunsaturated fatty acids: have 2 or more C=C double bonds.
More double bonds usually means a lower melting point.
Exam link:
Oils tend to contain more unsaturated fatty acids and are common in plants.
Fats tend to contain more saturated fatty acids and are common in endotherms.
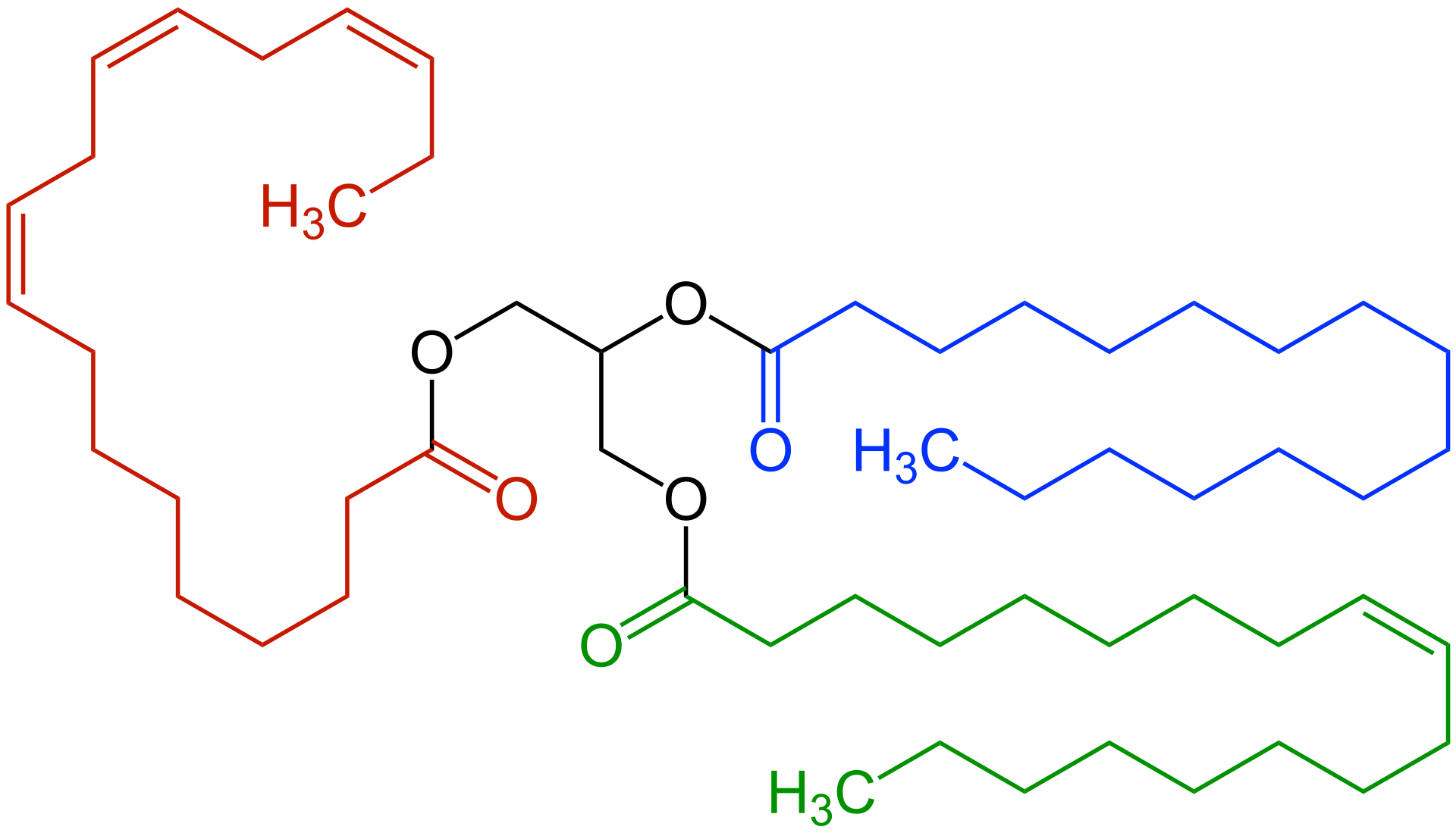
This diagram shows a triglyceride with glycerol linked to three fatty acid chains, including unsaturated chains with double bonds. It is useful for spotting the ester-linked structure of triglycerides and connecting double bonds with changes in physical properties such as melting point. Source
Phospholipids and membranes
Phospholipids form by condensation reactions.
One glycerol joins to 2 fatty acids and 1 phosphate group.
Phospholipids are amphipathic:
Hydrophilic phosphate head.
Hydrophobic fatty acid tails.
Because they have both hydrophilic and hydrophobic regions, phospholipids form bilayers in water.
This is the basis of cell membranes.
Exam wording to use: phospholipid bilayer forms as a consequence of hydrophilic and hydrophobic regions.
Strong link to later topic: phospholipid bilayers create a barrier between aqueous environments.
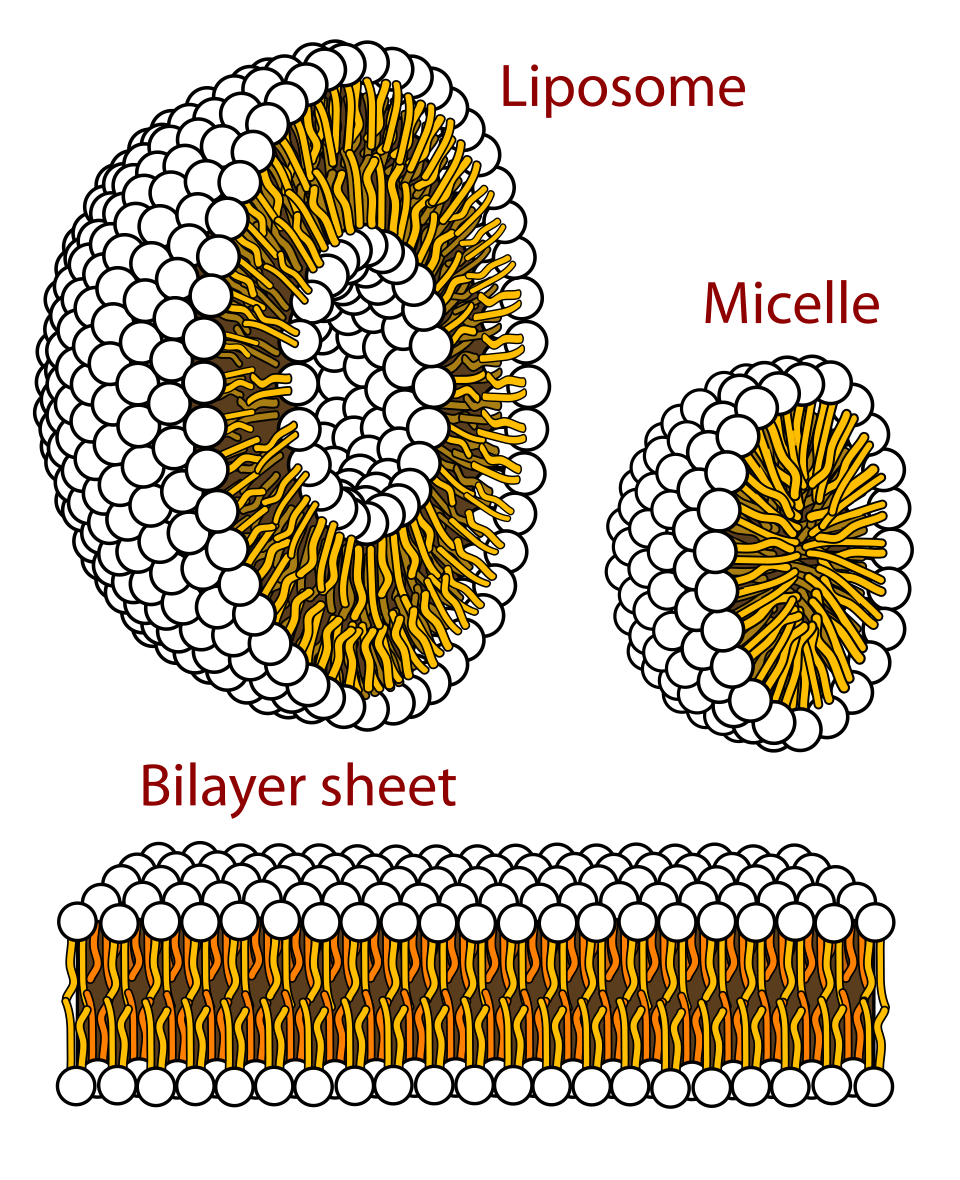
This diagram shows how phospholipids arrange themselves in water, including the bilayer arrangement relevant to cell membranes. It clearly shows hydrophilic heads facing water and hydrophobic tails away from water, which is the key IB idea. Source
Steroids
Steroids are non-polar lipids.
Because they are non-polar, they can pass through the phospholipid bilayer.
Required examples: oestradiol and testosterone.
Students should be able to identify a steroid from a molecular diagram.
Fast recognition tip: steroids usually show four fused carbon rings.
Checklist: can you do this?
Identify monosaccharides, triglycerides, phospholipids, and steroids from molecular diagrams.
Compare starch, glycogen, and cellulose in terms of monomer type, structure, and function.
Explain why triglycerides are good for energy storage and thermal insulation.
Explain why phospholipids form bilayers and why this matters biologically.
Relate structure to function using key phrases such as branching, insolubility, hydrogen bonding, hydrophobic, and amphipathic.
Exam traps to avoid
Do not confuse α-glucose with β-glucose.
Do not say cellulose is for energy storage; it is structural.
Do not forget that glycogen is animal storage and starch is plant storage.
Do not describe lipids as completely insoluble in water; they are sparingly soluble in aqueous solvents.
Do not forget the word amphipathic for phospholipids.
Ultra-compact comparisons
Glucose = small, soluble, transportable, immediate energy source.
Starch = plant storage polysaccharide.
Glycogen = animal storage polysaccharide, highly branched.
Cellulose = plant structural polysaccharide, β-glucose, straight chains, H-bonded.
Triglyceride = glycerol + 3 fatty acids, long-term energy store, insulation.
Phospholipid = glycerol + 2 fatty acids + phosphate, amphipathic, forms bilayers.
Steroid = non-polar lipid, passes through membranes, often recognized by 4 fused rings.

Shubhi is a seasoned educational specialist with a sharp focus on IB, A-level, GCSE, AP, and MCAT sciences. With 6+ years of expertise, she excels in advanced curriculum guidance and creating precise educational resources, ensuring expert instruction and deep student comprehension of complex science concepts.
Shubhi is a seasoned educational specialist with a sharp focus on IB, A-level, GCSE, AP, and MCAT sciences. With 6+ years of expertise, she excels in advanced curriculum guidance and creating precise educational resources, ensuring expert instruction and deep student comprehension of complex science concepts.