Enzymes as catalysts
Enzymes are biological catalysts that increase the rate of reaction without being used up.
Faster reactions are essential because cell reactions would otherwise be too slow to sustain life.
Enzymes allow reactions to occur at normal cellular temperatures rather than requiring extreme conditions.
Enzymes are central to metabolism, because each step in a pathway is usually controlled by a specific enzyme.
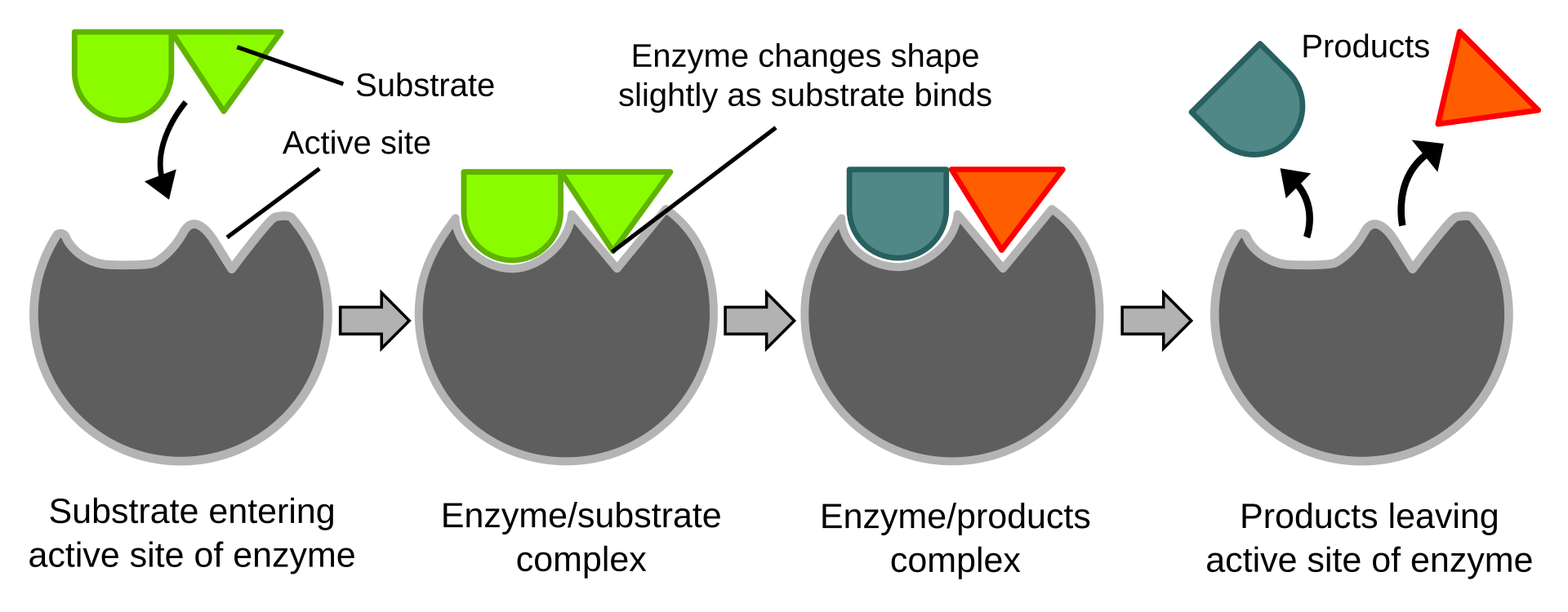
This diagram shows the induced-fit model: the active site changes shape slightly as the substrate binds, improving fit and catalysis. It is useful for explaining why enzyme action depends on both specificity and conformational change. Source
Metabolism: anabolic and catabolic pathways
Metabolism = the complex network of interdependent and interacting chemical reactions in living organisms.
Anabolic reactions build larger molecules from smaller ones and usually require energy.
Examples of anabolism: protein synthesis, glycogen formation, photosynthesis, and other condensation reactions.
Catabolic reactions break larger molecules into smaller ones and often release energy.
Examples of catabolism: digestion by hydrolysis and oxidation of substrates in respiration.
Metabolic pathways can be linear or cyclical; products of one reaction often become substrates for the next.
Because pathways are interdependent, changing one enzyme can affect the whole pathway.
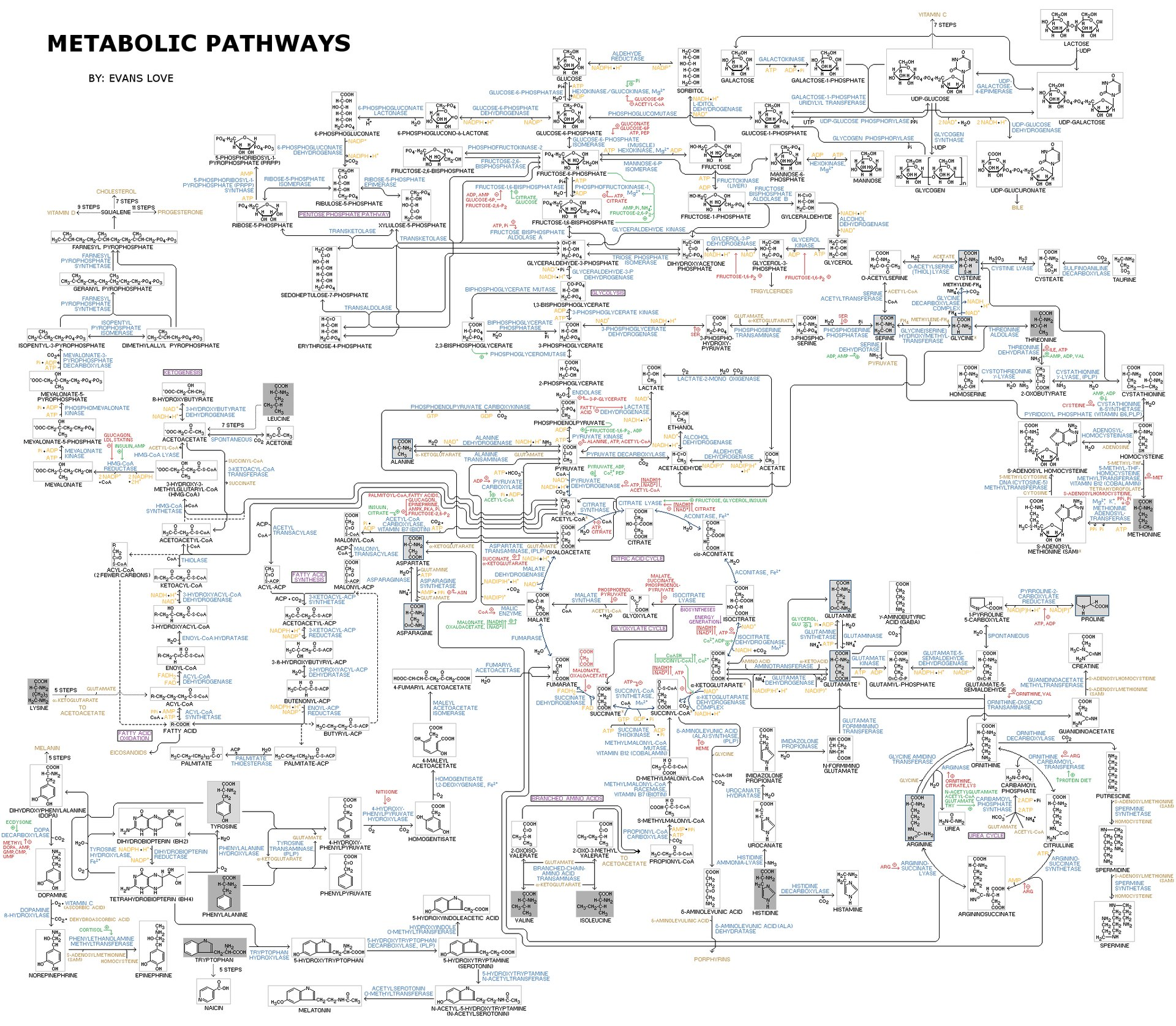
This image shows metabolism as an interconnected network rather than isolated reactions. It helps visualize how anabolic and catabolic pathways link together through shared intermediates. Source
Structure of enzymes and the active site
Enzymes are usually globular proteins with a specific three-dimensional shape.
The active site is the region where the substrate binds and where catalysis occurs.
The active site is formed by only a few amino acids, but its properties depend on the enzyme’s overall tertiary structure.
The shape and chemistry of the active site determine enzyme–substrate specificity.
Specificity means an enzyme only binds one substrate, or a small group of closely related substrates.
Induced fit, collisions and specificity
Enzyme action depends on molecular motion and successful collisions between substrate and active site.
A reaction occurs only when molecules collide with the correct orientation and enough energy.
In induced-fit binding, both the enzyme and the substrate change shape slightly when binding occurs.
This improves contact between substrate and active site and helps position bonds for reaction.
Some large substrates may be immobilized, while some enzymes may be immobilized in membranes.
Why denaturation matters
Denaturation = a change in enzyme three-dimensional structure that alters the active site.
If the active site changes shape, the substrate may no longer bind effectively.
Therefore, denaturation reduces or stops enzyme activity.
Denaturation links protein structure directly to enzyme function.
Factors affecting enzyme activity
Temperature, pH and substrate concentration all affect the rate of enzyme activity.
At low temperature, molecules move more slowly, so there are fewer successful collisions.
As temperature rises, kinetic energy increases, so collision frequency and reaction rate increase.
Above the optimum temperature, the enzyme begins to denature, so the rate falls rapidly.
Each enzyme has an optimum pH at which the active site has the correct shape and charge.
pH changes can alter ionic charges and bonding within the enzyme, distorting the active site.
As substrate concentration increases, rate rises because more active sites are occupied.
Eventually the rate plateaus because all active sites are occupied; the enzyme is saturated.
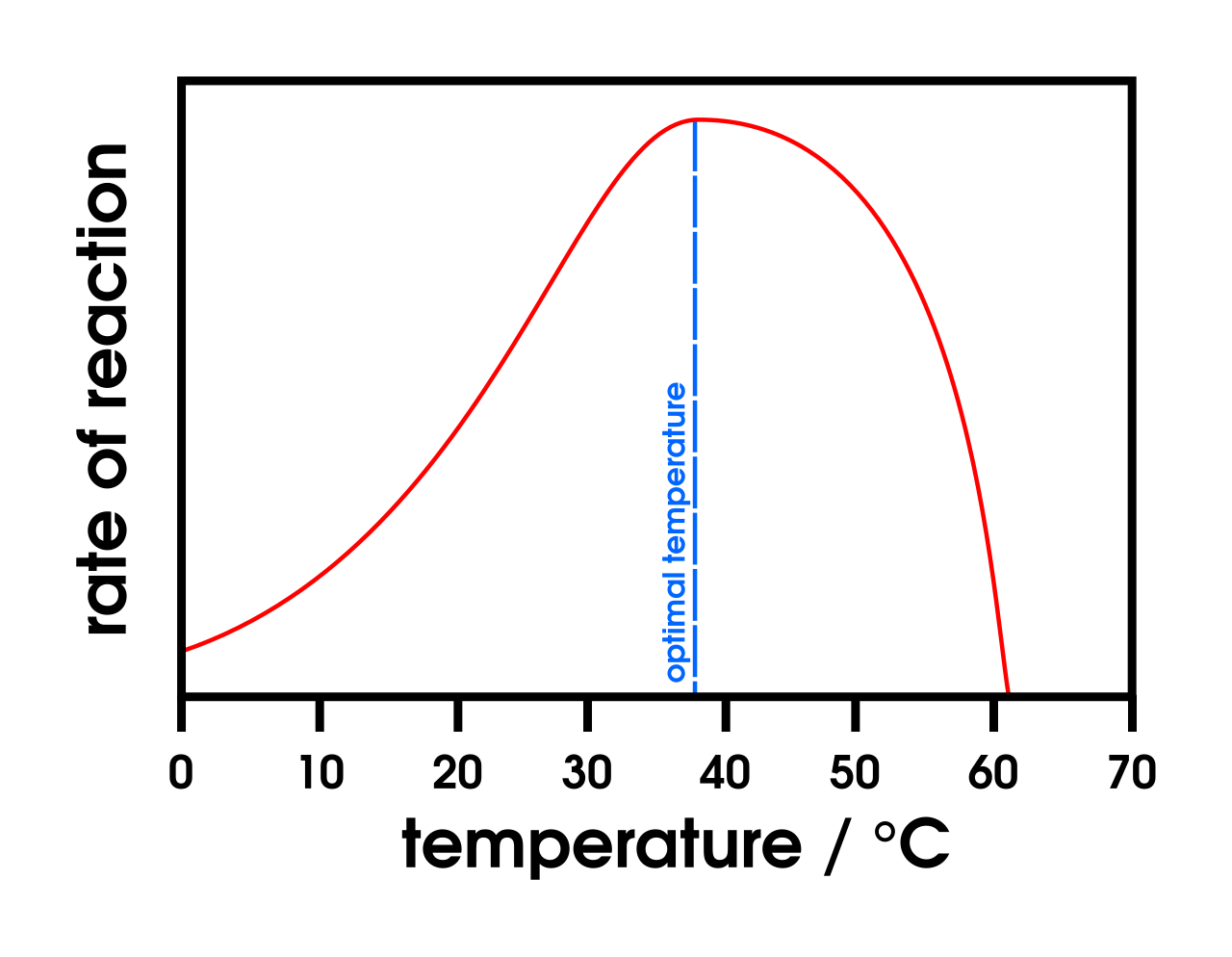
This graph shows the typical enzyme rate curve for temperature: increasing rate up to an optimum, then a sharp fall as denaturation occurs. It is ideal for explaining the role of collision frequency and why the drop after the optimum is steep. Source
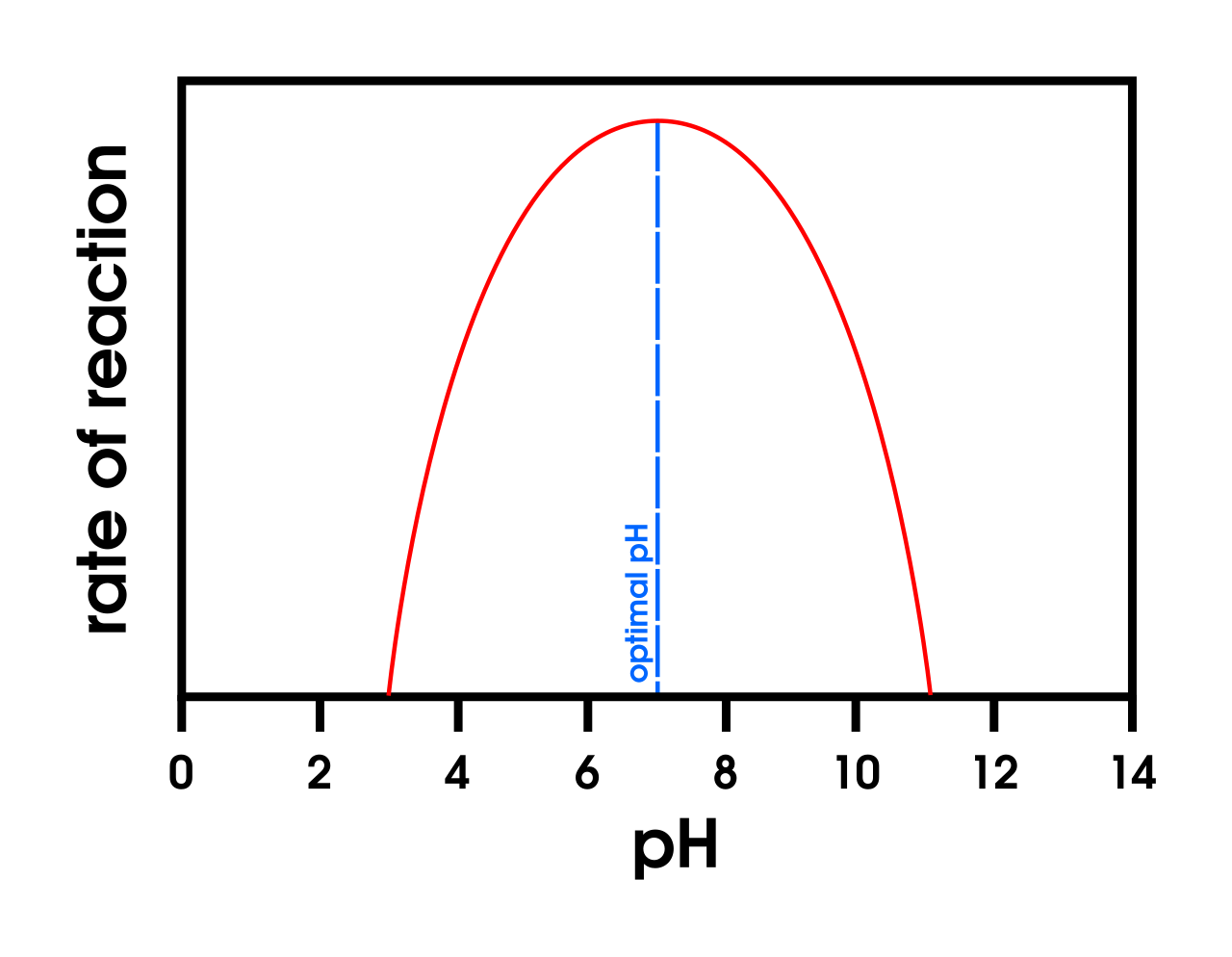
This graph shows the effect of pH on enzyme activity, with a clear optimum pH and lower activity on either side. It helps students interpret how changes in pH alter the active site and reduce catalysis. Source
Measuring enzyme-catalysed reactions
Enzyme activity is measured by determining reaction rate.
Reaction rate can be found by measuring substrate used up or product formed per unit time.
In practicals, students may use experimental data or secondary data to calculate rates.
Common graph skill: identify initial rate, compare conditions, and explain trends using collision theory and denaturation.
Be ready to interpret graphs for temperature, pH, substrate concentration, and activation energy.
Activation energy
Reactions require an initial input of energy called activation energy.
Enzymes lower activation energy, so more substrate molecules can react at a given temperature.
Enzymes do not change the overall energy of reactants or products.
In exam graphs, the enzyme-catalysed pathway has a lower peak than the uncatalysed pathway.
Energy is needed to break bonds in substrates, and energy is released when new bonds form in products.
HL only: intracellular vs extracellular enzyme action
Intracellular enzyme-catalysed reactions happen inside cells.
Examples: glycolysis and the Krebs cycle.
Extracellular enzyme-catalysed reactions happen outside cells.
Example: chemical digestion in the gut.
The location of enzyme action is linked to the function of the pathway.
HL only: heat production in metabolism
Metabolic reactions are not 100% efficient in energy transfer.
Therefore, some energy is always released as heat.
Mammals, birds, and some other animals use this heat production to help maintain a constant body temperature.
HL only: linear, cyclical and regulated pathways
Metabolic pathways may be linear or cyclical.
Glycolysis is a linear pathway.
The Krebs cycle and Calvin cycle are cyclical pathways.
Regulation of a pathway often occurs at a key enzyme-controlled step.
HL only: enzyme inhibition
Competitive inhibition happens when an inhibitor binds reversibly to the active site.
The inhibitor competes with the substrate for the active site.
Increasing substrate concentration can reduce the effect of competitive inhibition.
Statins are an example of competitive inhibitors.
Non-competitive inhibition happens when an inhibitor binds to an allosteric site.
The allosteric site is separate from the active site.
Binding causes a conformational change that alters the active site, preventing catalysis.
Increasing substrate concentration does not fully overcome non-competitive inhibition.
Binding at allosteric sites is specific and reversible in non-competitive inhibition.
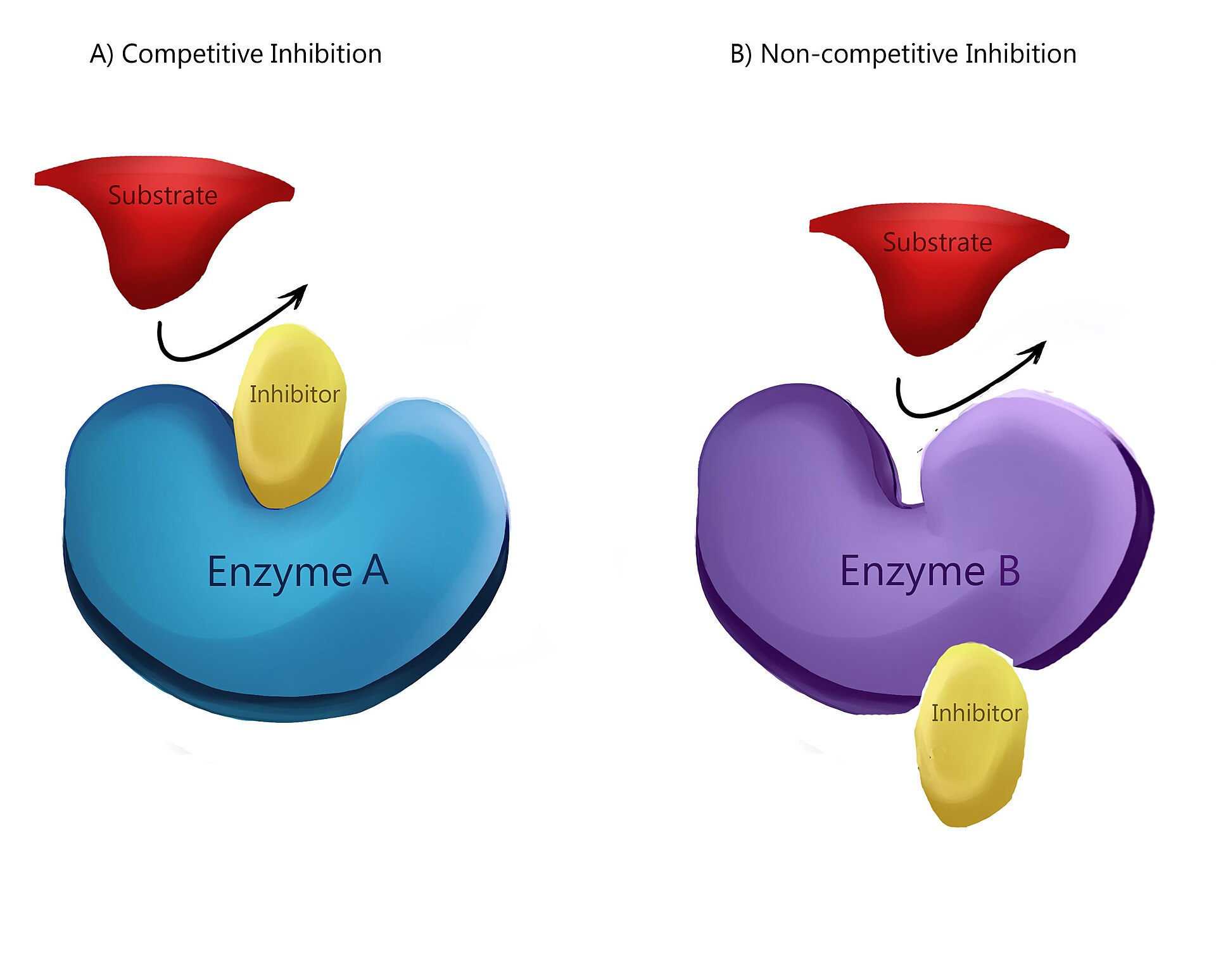
This image compares competitive and non-competitive inhibition. It clearly shows whether the inhibitor binds at the active site or at a separate allosteric site, which is a common exam comparison. Source
HL only: feedback inhibition and irreversible inhibition
Feedback inhibition regulates metabolic pathways by using the end product to inhibit an earlier enzyme in the pathway.
This prevents overproduction and helps maintain metabolic balance.
An example is the pathway producing isoleucine, where the end product inhibits an earlier step.
Mechanism-based inhibition is caused by irreversible binding of an inhibitor.
Irreversible binding causes chemical changes to the active site.
Penicillin is an example of mechanism-based inhibition.
Resistance to penicillin can occur when transpeptidases change so penicillin binds less effectively.
Exam graph skills
For temperature graphs: explain rising rate by increased kinetic energy/collisions, then falling rate by denaturation.
For pH graphs: explain deviations from the optimum by changes to bonding/charges and therefore the active site.
For substrate concentration graphs: explain the plateau using enzyme saturation and all active sites occupied.
For activation energy graphs: identify the lower activation energy in the catalysed reaction.
In all explanations, link back to active site shape, specificity, collisions, and protein structure.
Checklist: can you do this?
Explain how enzymes act as catalysts and why they are essential in metabolism.
Interpret graphs showing effects of temperature, pH, substrate concentration, and activation energy.
Distinguish clearly between anabolism and catabolism using correct examples.
Compare competitive, non-competitive, and feedback inhibition.
Calculate or describe reaction rate from practical data for an enzyme-catalysed reaction.

Shubhi is a seasoned educational specialist with a sharp focus on IB, A-level, GCSE, AP, and MCAT sciences. With 6+ years of expertise, she excels in advanced curriculum guidance and creating precise educational resources, ensuring expert instruction and deep student comprehension of complex science concepts.
Shubhi is a seasoned educational specialist with a sharp focus on IB, A-level, GCSE, AP, and MCAT sciences. With 6+ years of expertise, she excels in advanced curriculum guidance and creating precise educational resources, ensuring expert instruction and deep student comprehension of complex science concepts.