Stratospheric ozone
· Guiding idea: the stratospheric ozone layer protects life by absorbing harmful ultraviolet (UV) radiation and is maintained by a steady-state equilibrium between ozone formation and ozone destruction.
· Exam distinction: ozone depletion is not a cause of global warming; it mainly increases UVB reaching Earth’s surface.
· Key human cause: ozone-depleting substances (ODSs), especially CFCs, release reactive halogens that catalyse ozone destruction.
· Key management example: the Montreal Protocol is a major case study of successful international cooperation.
Electromagnetic radiation and UV risk
· The Sun emits electromagnetic radiation across a spectrum: radio waves → microwaves → infrared → visible light → ultraviolet → X-rays → gamma rays.
· Shorter wavelength = higher frequency = higher energy, so UV radiation is more biologically damaging than visible or infrared radiation.
· UVA has the longest UV wavelength and mostly reaches the surface; UVB is partly absorbed but some reaches the surface; UVC has the shortest wavelength and is fully absorbed by stratospheric ozone.
· UVB and UVC are most exam-relevant because they are high-energy and can damage living tissue.
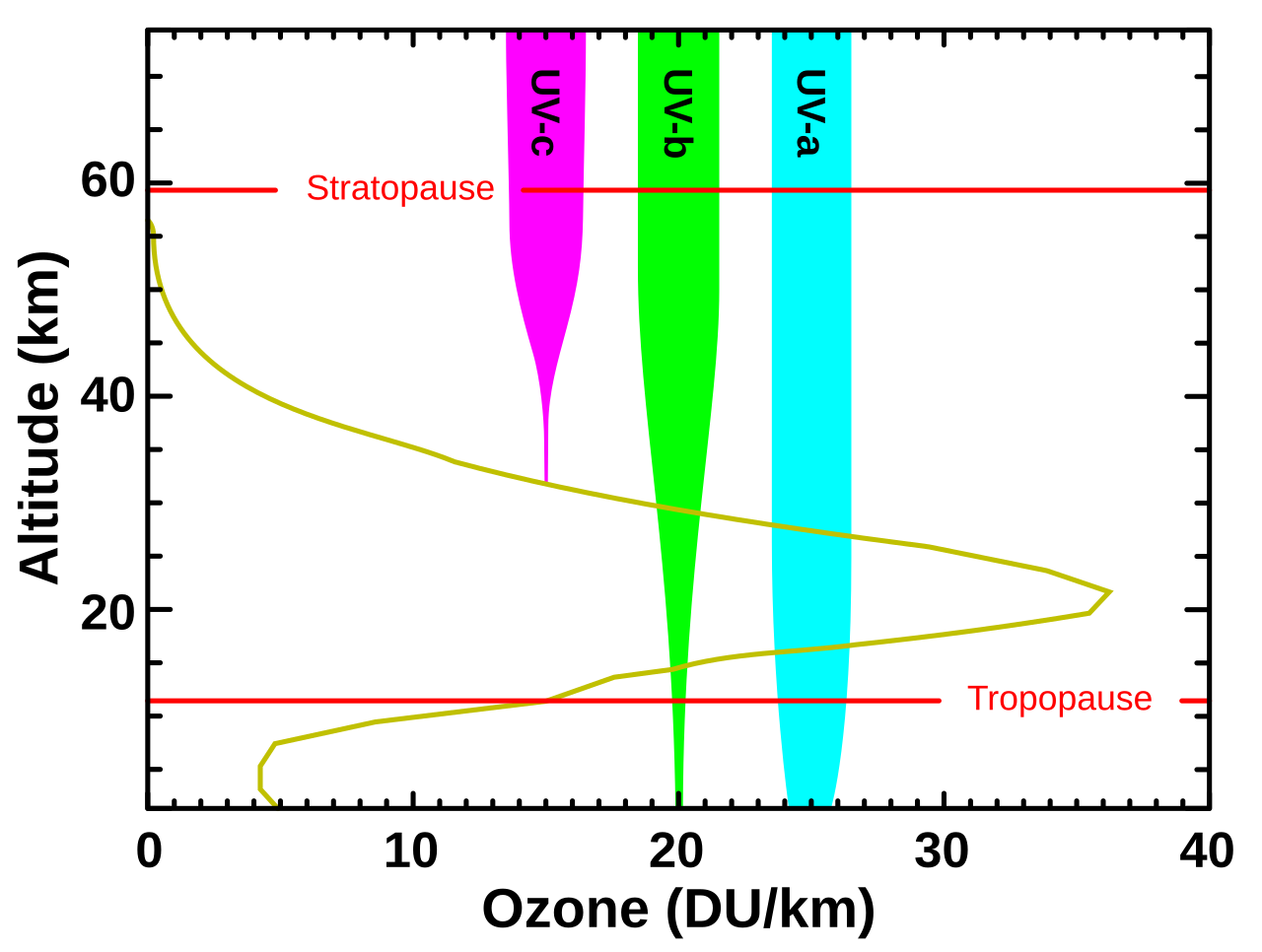
This graph shows that stratospheric ozone absorbs all UVC and much of UVB, while most UVA reaches Earth’s surface. It is useful for explaining why ozone depletion mainly increases biologically harmful UVB exposure. Source
Role of stratospheric ozone
· Stratospheric ozone absorbs UV radiation, reducing the amount reaching Earth’s surface.
· It absorbs all incident UVC and most UVB, protecting living organisms from harmful radiation.
· This protection helps maintain primary productivity, DNA stability, and human health.
· Tropospheric ozone is different: it is a harmful air pollutant linked to photochemical smog, whereas stratospheric ozone is protective.
Effects of increased UV radiation
· UV radiation damages DNA, causing mutations that can lead to cancer.
· In humans, increased UV exposure can cause sunburn, premature skin ageing, skin cancer, and cataracts.
· In ecosystems, increased UV can reduce photosynthesis in phytoplankton, lowering primary productivity and disrupting aquatic food webs.
· Exam data may show relationships between ozone concentration, UVB exposure, skin cancer/cataracts, or phytoplankton productivity.
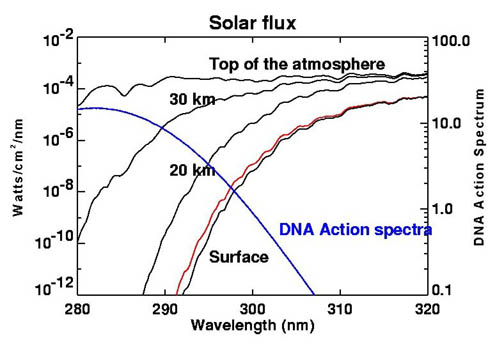
This image links ozone absorption to biological damage by showing that ozone strongly absorbs the UV wavelengths most likely to damage DNA. It is useful for explaining why ozone depletion increases health and ecosystem risks. Source
Natural ozone equilibrium
· Ozone concentration stays relatively constant over long periods because of a steady-state equilibrium.
· Ozone formation and ozone destruction occur continuously and naturally in the stratosphere.
· A steady-state equilibrium means flows continue, but the overall storage of ozone remains stable when formation rate equals destruction rate.
· If destruction exceeds formation, the equilibrium shifts towards ozone depletion.
· SL students do not need chemical equations for this equilibrium, but must understand the systems idea.
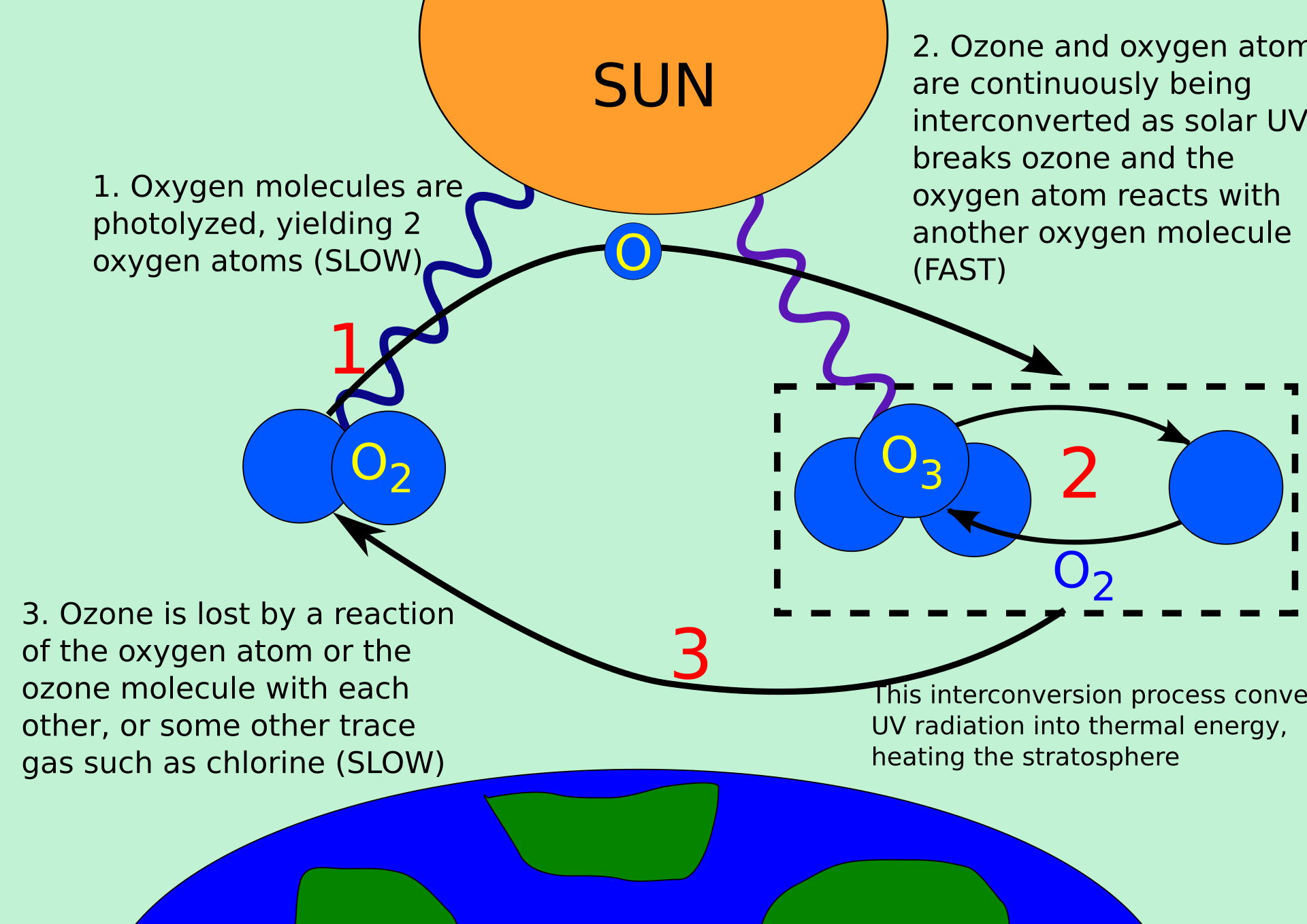
This diagram illustrates the natural cycling between oxygen and ozone in the stratosphere. It supports the idea of a steady-state equilibrium between ozone formation and destruction. Source
Ozone-depleting substances (ODSs)
· ODSs are substances that destroy ozone molecules and increase the natural breakdown rate of ozone.
· Major examples include chlorofluorocarbons (CFCs) and other halogen-containing compounds.
· CFCs were widely used in aerosols, refrigerators, and air conditioning systems because they were stable and useful as coolants/propellants.
· Their stability allows them to persist long enough to reach the stratosphere, where UV breaks them down and releases reactive chlorine.
· A single chlorine radical can destroy many ozone molecules because it acts as a catalyst and is regenerated.
Ozone depletion and the ozone hole
· Ozone depletion means a reduction in the concentration of stratospheric ozone.
· Depletion allows more UVB radiation to reach Earth’s surface, increasing risks to ecosystems and human health.
· Ozone depletion has affected the stratosphere globally, but is most severe over the poles.
· The Antarctic ozone hole forms each spring due to the combination of ODSs, polar atmospheric conditions, and seasonal chemistry.
· The ozone hole is not a literal empty hole; it is an area of very low ozone concentration.
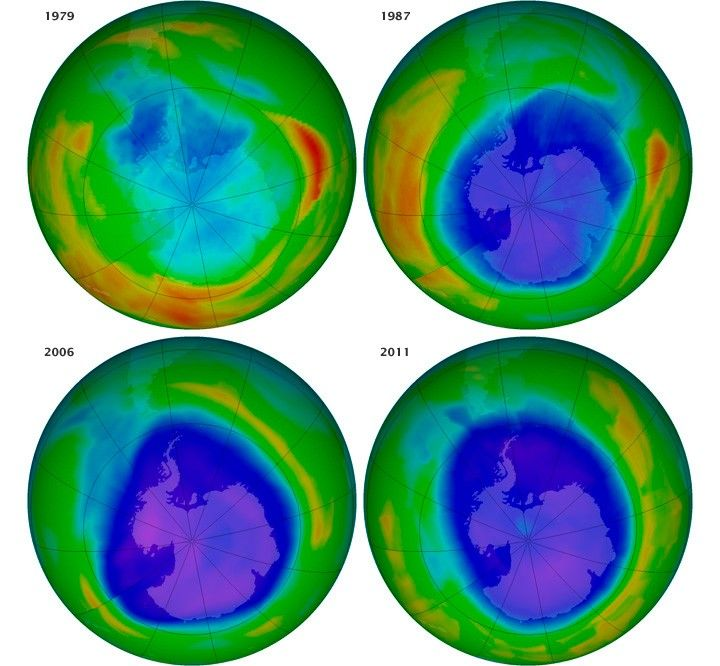
This NASA visual shows how satellite data are used to monitor the Antarctic ozone hole over time. It is useful evidence for evaluating whether international action has reduced ozone depletion. Source
Montreal Protocol and management
· The Montreal Protocol is an international treaty regulating the production, trade and use of CFCs and other ODSs.
· It is considered a highly successful example of global environmental cooperation.
· Success factors include clear scientific evidence, global participation, legally binding controls, phase-out targets, and available substitutes.
· The treaty helped reduce emissions of many ODSs and supported recovery of the ozone layer.
· It is an important comparison point for other global issues requiring cooperation, such as climate change.
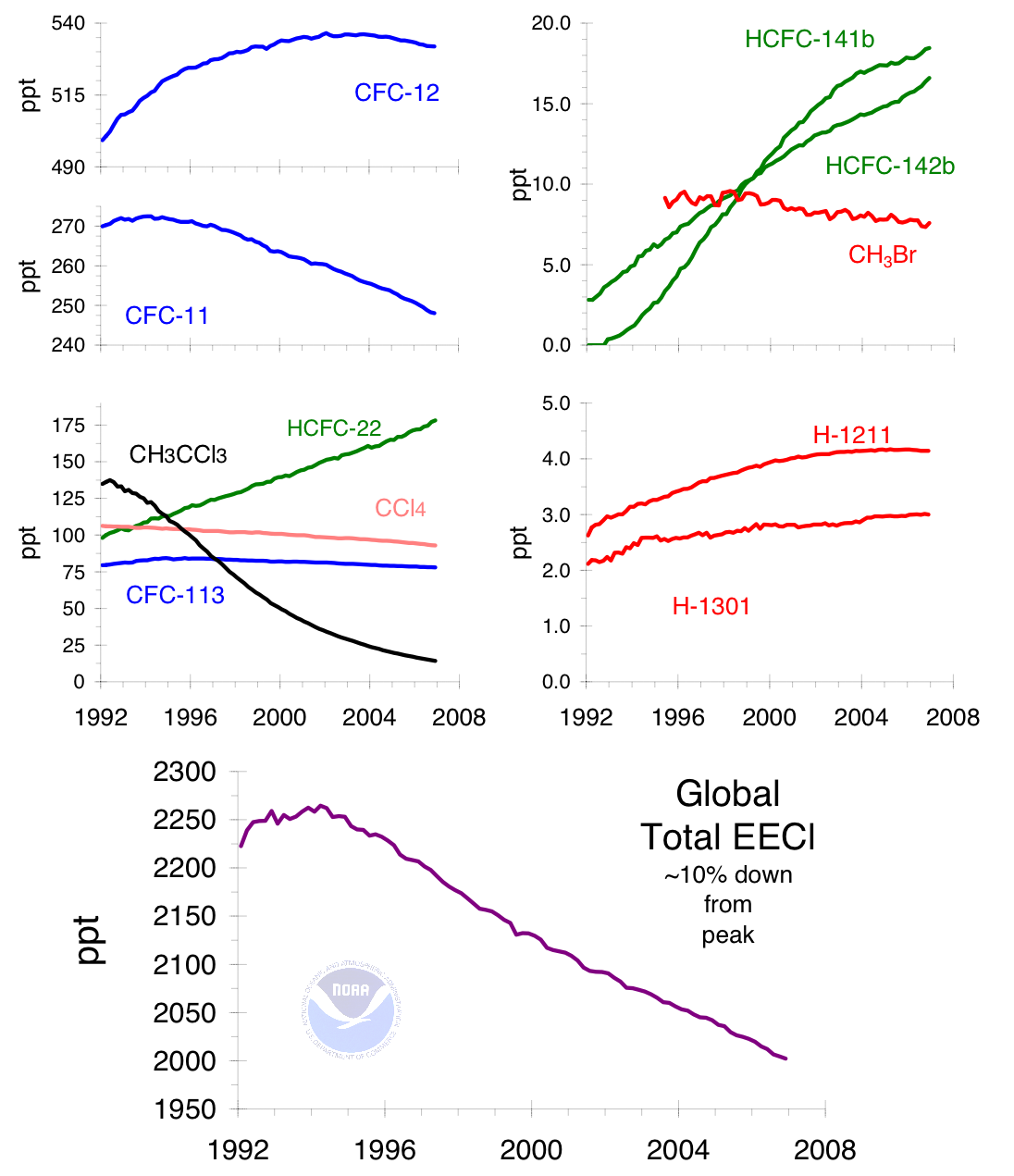
This graph shows how the atmospheric influence of controlled ozone-depleting gases changed after international regulation. It is useful for linking the Montreal Protocol to changes in ozone-depleting chemical concentrations. Source
Planetary boundary link
· Stratospheric ozone depletion is one of the planetary boundaries.
· The syllabus emphasises that action under the Montreal Protocol has prevented this boundary from being crossed.
· Evidence can include ozone hole area over time, ozone concentration data, and trends in CFC/ODS concentrations.
· Strong exam answers connect science → policy → environmental recovery.
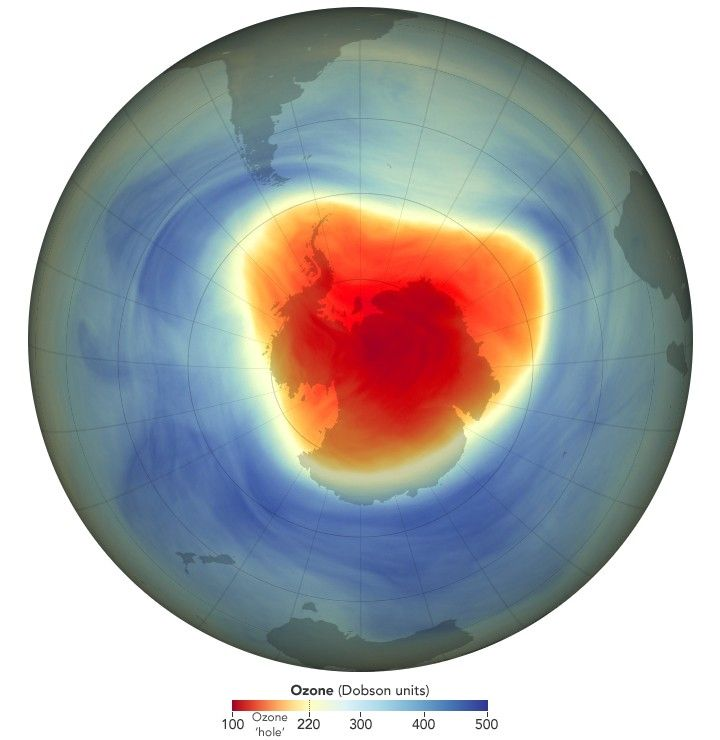
This map provides recent evidence that the Antarctic ozone hole continues to be monitored using satellite data. It supports evaluation of long-term ozone recovery following controls on ozone-depleting chemicals. Source
HL only: ozone formation and destruction equations
· HL only: chemical equations are required for the formation and destruction of ozone.
· Oxygen photolysis:
· Ozone formation:
· Ozone photolysis:
· Natural equilibrium: ozone is continuously formed and destroyed, maintaining a steady-state concentration when rates are balanced.
HL only: catalytic ozone destruction by halogens
· HL only: ODSs release halogens such as chlorine and fluorine into the stratosphere, causing catalytic ozone breakdown.
· Example chlorine mechanism:
· Regeneration of chlorine:
· Overall reaction:
· Because chlorine is regenerated, it can destroy many ozone molecules before being removed from the stratosphere.
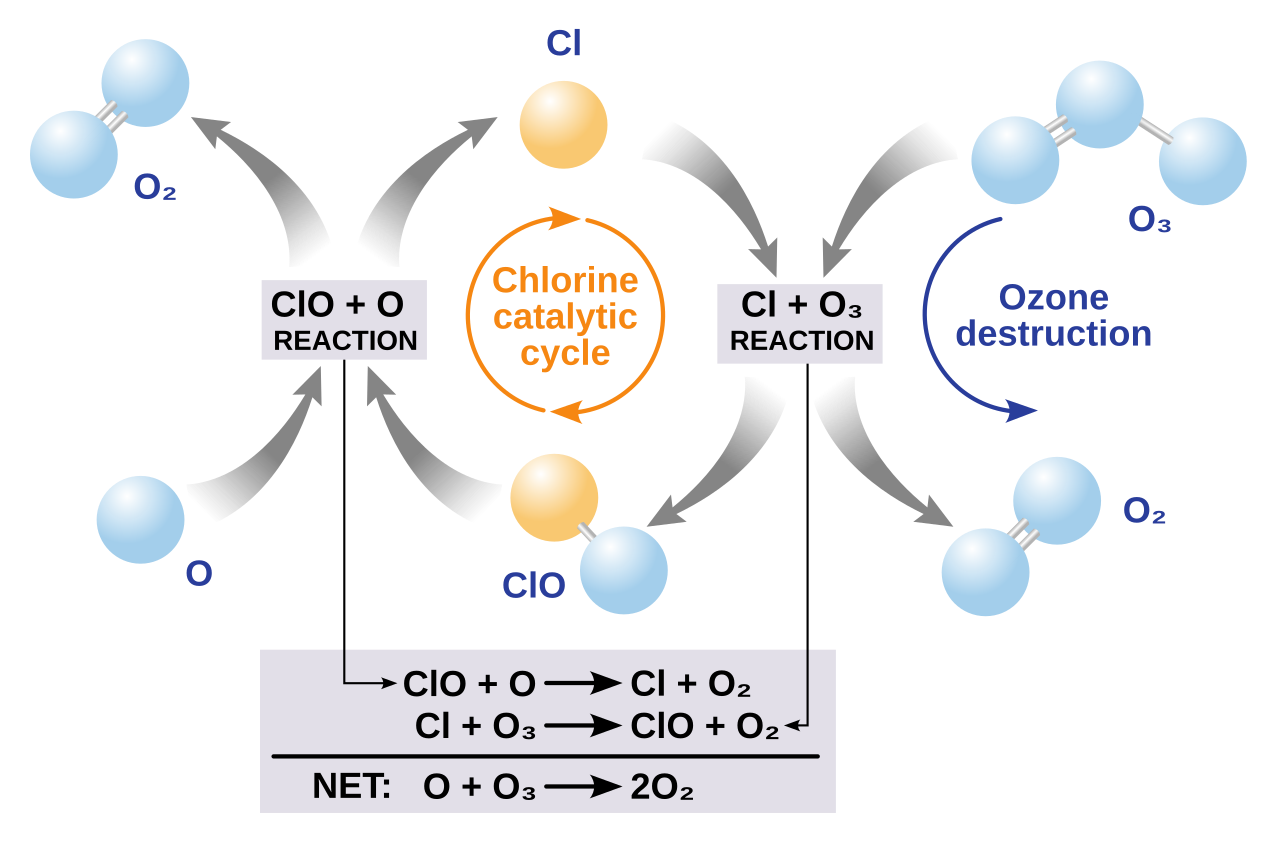
This diagram shows how chlorine is regenerated during ozone destruction, making it a catalyst. It is especially useful for HL students learning the ozone destruction equations. Source
HL only: polar ozone depletion
· HL only: polar ozone depletion is strongest in spring because of unique chemical and atmospheric conditions in the polar stratosphere.
· Polar stratospheric clouds (PSCs) provide active surfaces that enhance ozone destruction reactions.
· Volcanic aerosols can also provide surfaces that enhance ozone-depleting reactions.
· When sunlight returns in spring, stored chlorine compounds become reactive and rapidly destroy ozone.
· This explains why the ozone hole is seasonal and especially severe over Antarctica.
HL only: HFCs, Kigali Amendment and air conditioning
· HL only: hydrofluorocarbons (HFCs) were developed to replace CFCs because they cause much less ozone depletion.
· Problem: many HFCs are potent greenhouse gases, so they contribute to climate change.
· The Kigali Amendment to the Montreal Protocol controls HFCs.
· Air conditioning units are important because they are energy-intensive, may leak refrigerants, and increase GHG emissions.
· Alternatives include better building design, passive cooling, urban greening, rewilding of cities, and careful refrigerant recovery from old appliances.
Common exam mistakes to avoid
· Do not say ozone depletion causes global warming; these are different atmospheric issues.
· Do not confuse stratospheric ozone with tropospheric ozone.
· Do not say the ozone hole is a physical gap; it is an area of reduced ozone concentration.
· Do not describe the Montreal Protocol vaguely; link it to CFC/ODS phase-out, international cooperation, and ozone recovery evidence.
· For HL, do not forget that chlorine is regenerated, which is why ozone destruction is catalytic.
Checklist: can you do this?
· Explain how stratospheric ozone protects life by absorbing UVC and most UVB.
· Describe steady-state equilibrium between ozone formation and ozone destruction.
· Explain how ODSs/CFCs shift equilibrium towards ozone depletion.
· Interpret data showing changes in UV exposure, ozone hole area, or ODS concentrations.
· Evaluate the Montreal Protocol as a successful example of international environmental management.

Shubhi is a seasoned educational specialist with a sharp focus on IB, A-level, GCSE, AP, and MCAT sciences. With 6+ years of expertise, she excels in advanced curriculum guidance and creating precise educational resources, ensuring expert instruction and deep student comprehension of complex science concepts.
Shubhi is a seasoned educational specialist with a sharp focus on IB, A-level, GCSE, AP, and MCAT sciences. With 6+ years of expertise, she excels in advanced curriculum guidance and creating precise educational resources, ensuring expert instruction and deep student comprehension of complex science concepts.