Urban air pollution
· Guiding question: How can urban air pollution be effectively managed?
· Urban air pollution = pollution caused by human inputs to atmospheric systems in built-up areas.
· Main pollutants to know: nitrogen oxides (NOx), sulfur dioxide (SO₂), carbon monoxide (CO) and particulate matter (PM).
· PM2.5 = fine particulate matter with diameter ≤ 2.5 micrometres.
· PM10 = larger particulate matter with diameter ≤ 10 micrometres.
· Exam focus: link source → pollutant → atmospheric process → impact → management strategy.
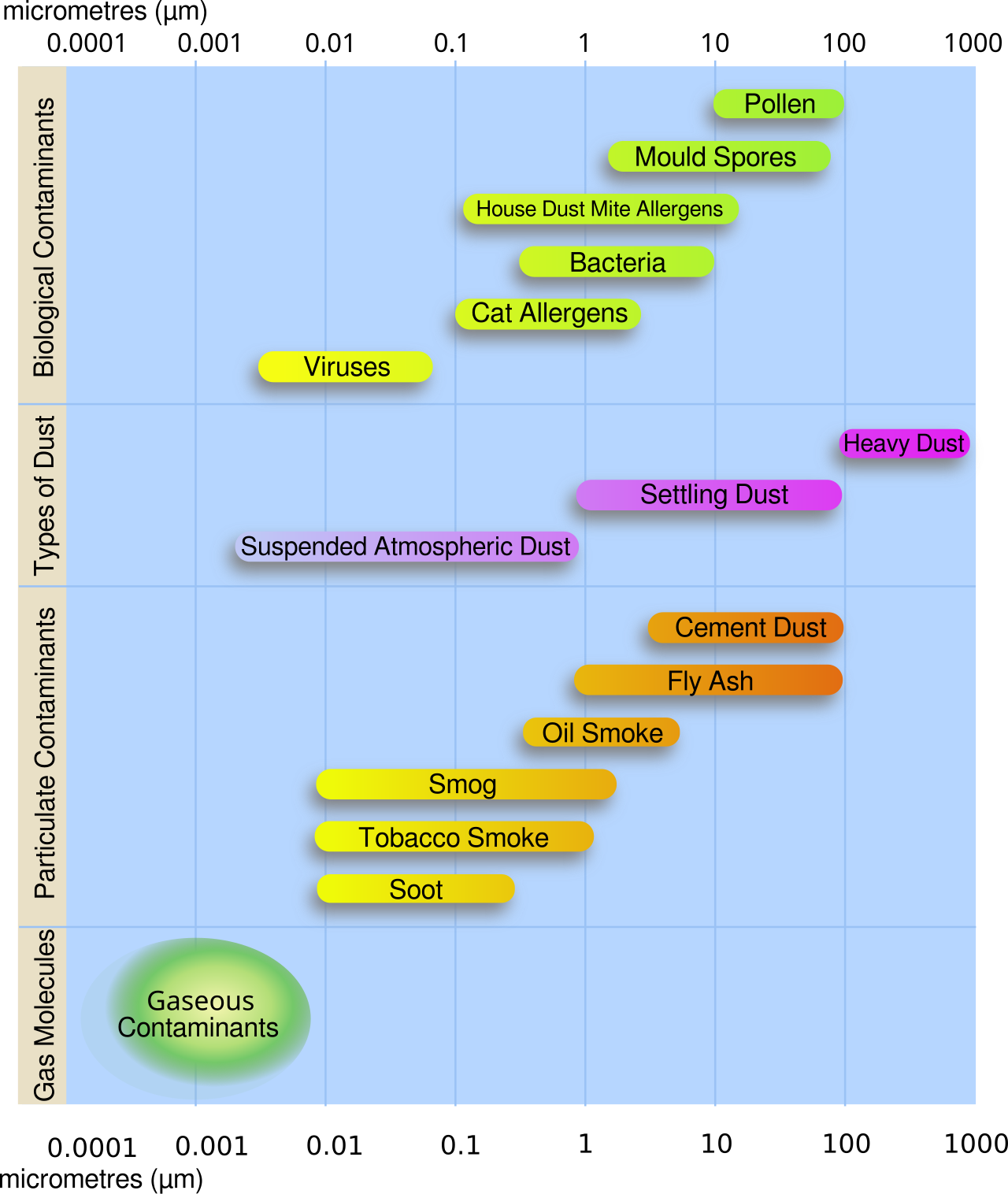
This diagram helps students visualise why PM2.5 is more dangerous than larger particles: fine particles can penetrate deeper into the respiratory system. It is useful beside the definitions of PM2.5 and PM10. Source
Primary pollutants and sources
· Primary pollutants = pollutants that are directly active at the point of emission.
· Natural sources: forest fires, dust, volcanic eruptions.
· Anthropogenic sources: burning fossil fuels, burning biomass, agricultural/forest clearance burning, energy production, construction dust, road dust.
· Most common urban pollutants are linked directly or indirectly to fossil fuel combustion.
· Key primary urban pollutants: PM2.5, PM10, carbon monoxide, sulfur dioxide.
· Key secondary urban pollutants: tropospheric ozone, nitric acid, sulfuric acid.
Managing urban air pollution
· Management aims to reduce pollutant production, control pollutants at release, or reduce exposure/restore damaged systems.
· Transport strategies: improved public transport, cycling infrastructure, limited car use, pedestrianized town centres.
· Urban greening strategies: trees, natural screens, green walls to intercept particulates and improve urban air quality.
· Technology/regulation: compulsory catalytic converters to reduce harmful vehicle emissions.
· Strong answers should evaluate strategies using scale, cost, effectiveness, equity, and long-term sustainability.
Acid rain formation
· Acid rain forms when NOx and sulfur dioxide react with water and oxygen in the atmosphere to form nitric acid and sulfuric acid.
· Sulfur dioxide pathway: then oxidation can form .
· Nitrogen oxide pathway: reacts in the atmosphere to form nitric acid, .
· Main acids in acid deposition: nitric acid and sulfuric acid.
· Acid deposition can occur as wet deposition or dry deposition.
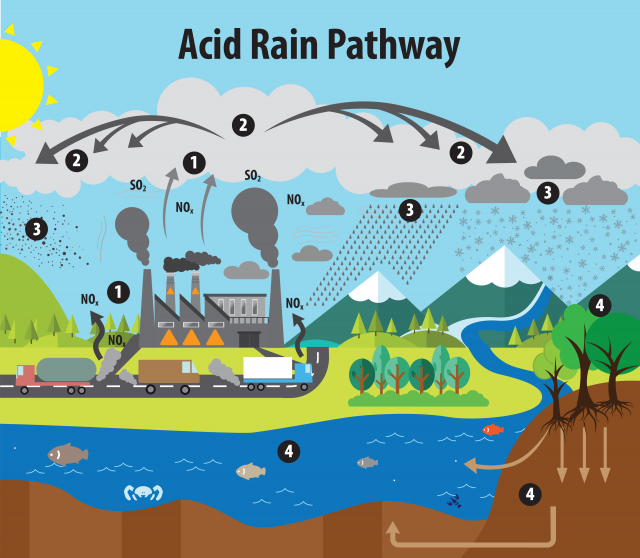
This diagram clearly links emissions, atmospheric transformation, transport, and deposition. It is useful for explaining acid rain as a systems process rather than a single event. Source
Acid rain impacts
· Terrestrial ecosystems: causes leaching of nutrients, toxification of soil, and direct damage to foliage.
· Freshwater ecosystems: increases toxicity through aluminium solubilization, damaging fish gills and invertebrate exoskeletons.
· Buildings/materials: corrodes marble, limestone, steel, paint, and other construction materials.
· Human health: nitrate and sulfate particles contribute to PM2.5, causing tissue damage and lung inflammation.
· Strong exam answers should separate ecological, human health, and built environment impacts.
Managing SO₂, NOx and acid rain impacts
· Alter human activity: switch to alternative energy sources and reduce fossil fuel use.
· Control at point of release: use scrubbers in power stations and catalytic converters in vehicles.
· Restore damaged systems: provide healthcare, add limestone to acidified lakes, and add fertilizer where nutrients have been leached.
· Management can be evaluated at three levels: prevention, emission control, and restoration.
· Best exam phrasing: source reduction is usually more sustainable than treating symptoms after pollution has occurred.
HL only: photochemical smog formation
· Photochemical smog forms when sunlight acts on primary pollutants, causing chemical transformation into secondary pollutants.
· Primary pollutants involved: NOx and volatile organic compounds (VOCs).
· Secondary pollutants formed: peroxyacyl nitrates (PANs) and tropospheric ozone.
· Tropospheric ozone is harmful at ground level, unlike protective stratospheric ozone.
· Photochemical smog is especially associated with urban traffic, sunny conditions, and poor dispersion of pollutants.
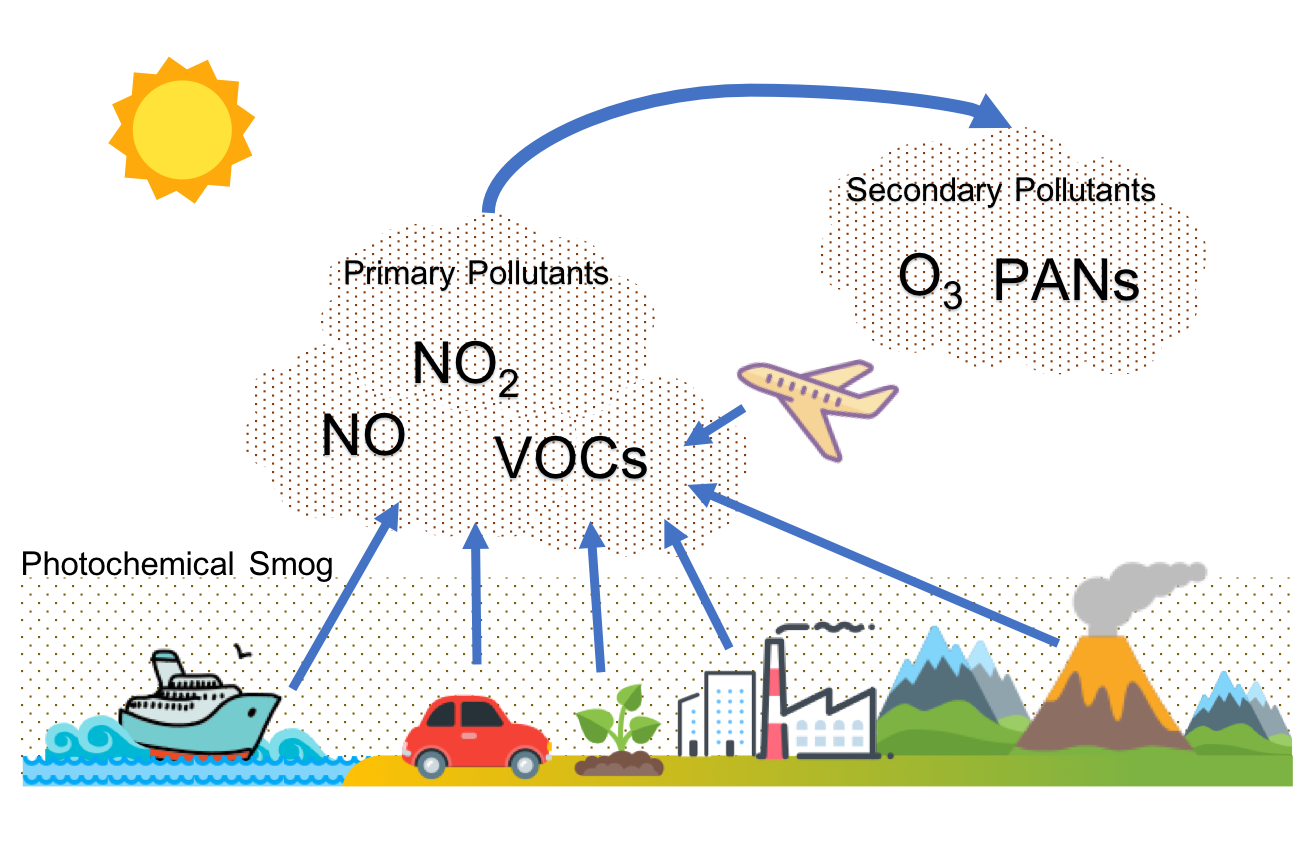
This diagram is useful for the HL section because it shows how NOx and VOCs act as precursors for secondary pollutants. It supports the key distinction between primary pollutants and secondary pollutants. Source
HL only: conditions that intensify photochemical smog
· Meteorological factors: abundant insolation, reduced wind, and temperature inversion.
· Topographical factors: pollution is intensified where cities are surrounded by mountains or high buildings.
· Temperature inversion = a layer of warmer air traps cooler polluted air close to the ground, reducing vertical mixing.
· Reduced wind lowers pollutant dispersal, so pollutants accumulate.
· High insolation increases photochemical reactions, raising tropospheric ozone and PAN formation.
HL only: tropospheric ozone impacts
· Direct biological impacts: damages plant cuticles and membranes, causes eye irritation in humans and mammals, and contributes to respiratory illness.
· Direct physical impacts: damages fabrics and rubber materials.
· Indirect societal impacts: increases pressure on healthcare systems and reduces economic output through illness and lost workforce productivity.
· Environmental justice link: poorer communities often carry a larger share of pollution impacts.
· Strong HL answers should link tropospheric ozone → health/ecological damage → economic and social costs.
Practical and data skills
· Plan an investigation using an indicator species as a correlate for local air pollution.
· Use graphs showing diurnal changes in urban air pollutants.
· Use secondary databases to study local air-quality change over time.
· Apply a suitable statistical tool to test whether observed air-quality change is significant.
· Interpret patterns such as rush-hour pollutant peaks, midday ozone formation, and reduced dispersion during temperature inversions.
Checklist: can you do this?
· Identify primary and secondary urban air pollutants and match each to its source.
· Explain how NOx and SO₂ form acid rain and describe key impacts.
· Compare management strategies for urban air pollution using prevention, control, and restoration.
· Interpret pollutant graphs showing diurnal changes and explain the causes of peaks.
· HL: explain how photochemical smog forms and why insolation, wind, temperature inversion, and topography intensify it.
Exam traps to avoid
· Do not confuse tropospheric ozone with stratospheric ozone: ground-level ozone is a pollutant, stratospheric ozone is protective.
· Do not call photochemical smog a primary pollutant: it is formed by secondary pollutants produced from primary pollutant reactions.
· Do not say PM10 is smaller than PM2.5: PM2.5 is the finer fraction.
· Do not only list strategies: explain how each strategy reduces emissions, exposure, or impacts.
· Do not ignore environmental justice: pollution impacts can be unevenly distributed across society.

Shubhi is a seasoned educational specialist with a sharp focus on IB, A-level, GCSE, AP, and MCAT sciences. With 6+ years of expertise, she excels in advanced curriculum guidance and creating precise educational resources, ensuring expert instruction and deep student comprehension of complex science concepts.
Shubhi is a seasoned educational specialist with a sharp focus on IB, A-level, GCSE, AP, and MCAT sciences. With 6+ years of expertise, she excels in advanced curriculum guidance and creating precise educational resources, ensuring expert instruction and deep student comprehension of complex science concepts.