Biogeochemical cycles: core idea
· Biogeochemical cycles keep chemical elements available to living organisms.
· Exam focus = how carbon and nitrogen move through systems, and how human activity disrupts those cycles.
· Always think in terms of a systems model: stores, sinks, sources, and flows.
· Link cycle disruption to ecosystem sustainability, planetary boundaries, and management strategies.
Key systems language
· Store / storage = place where matter is held.
· Sink = place with net accumulation of an element.
· Source = place with net release of an element.
· Residence time = average time an atom remains in a store.
· Transfer = movement of matter from one place to another without changing chemical form.
· Transformation = process that changes chemical nature/state of matter.
· In exam answers, identify whether a named process is a transfer or a transformation.
Carbon cycle: stores and flows
· Organic carbon stores = organisms, crude oil, natural gas.
· Inorganic carbon stores = atmosphere, soils, oceans.
· Major carbon flows in ecosystems: photosynthesis, feeding, defecation, cellular respiration, death, decomposition.
· Photosynthesis removes CO₂ from the atmosphere and stores carbon in biomass.
· Cellular respiration returns carbon to the atmosphere/water as CO₂.
· Decomposition recycles carbon from dead organic matter.
· Carbon sequestration = capture of atmospheric/gaseous CO₂ and storage in solid or liquid form.
· Trees naturally sequester carbon by converting CO₂ into biomass.
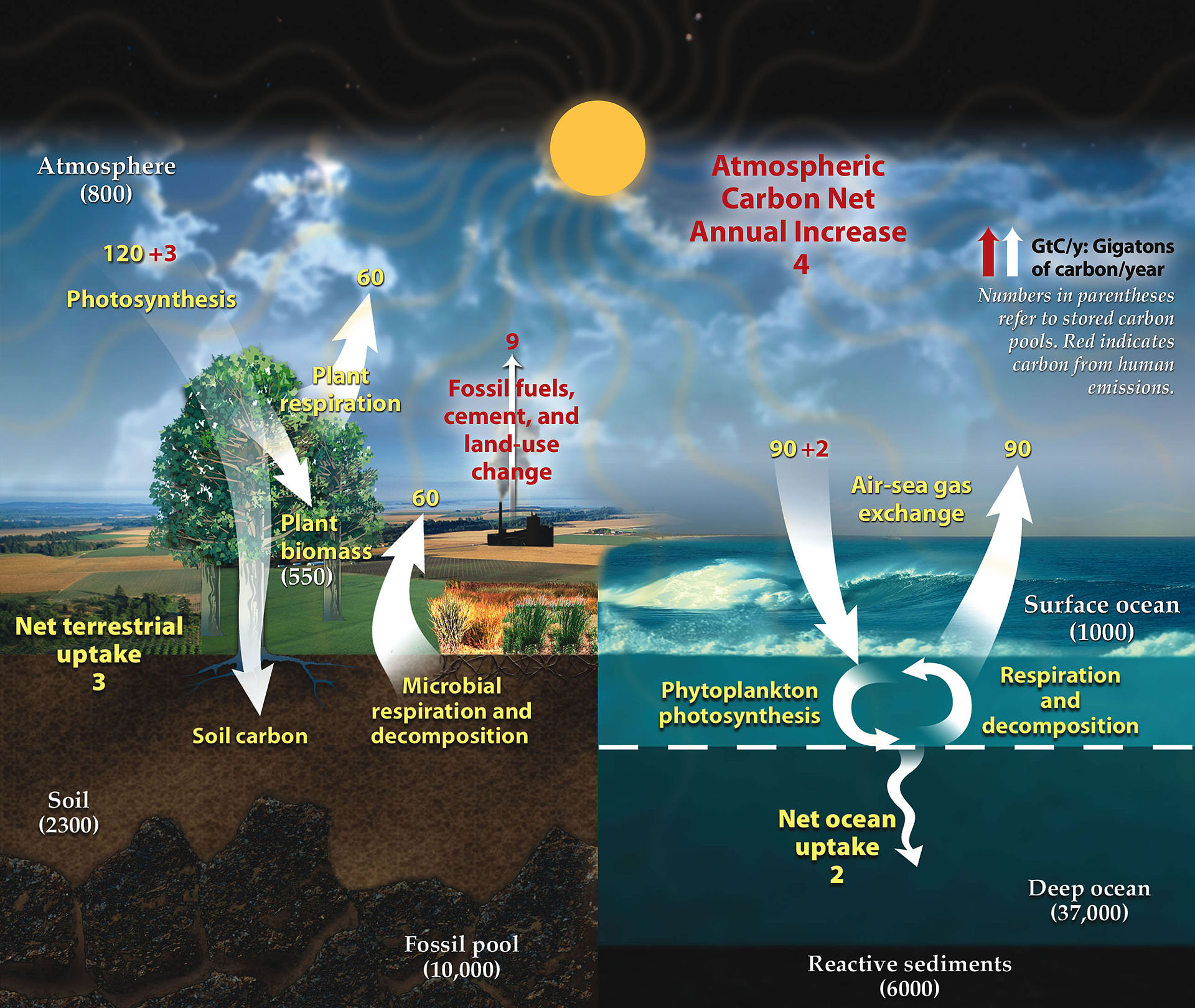
This diagram shows the major carbon stores and flows between atmosphere, plants, animals, soils, oceans, and fossil fuels. It is useful for identifying where carbon is stored, where it is transferred, and how human combustion changes the cycle. Use it to practise explaining pathways in full sentences. Source
Carbon stores, sinks and sources in ecosystems
· A system is a carbon sink when inputs exceed outputs.
· A system is a carbon source when outputs exceed inputs.
· A system is a carbon store when it is roughly in equilibrium over time.
· Young forest = usually a sink because photosynthesis > respiration.
· Mature forest = usually a store because uptake and release are more balanced.
· Burned or deforested forest = source because stored carbon is released.
· Fossil fuels are very long-term carbon stores, but become major sources when burned.
· In essays, always compare rate of uptake versus rate of release.
Carbon cycle and agriculture
· Agricultural systems can act as carbon stores, sinks, or sources depending on management.
· Practices that can increase soil carbon storage: crop rotation, cover crops, no-till farming.
· Practices that can increase carbon release: wetland drainage, monoculture, heavy tillage.
· Longer-term cropping systems such as timber production may store carbon in both biomass and harvested products.
· Exam theme: agriculture can either support or reduce sustainability depending on technique.
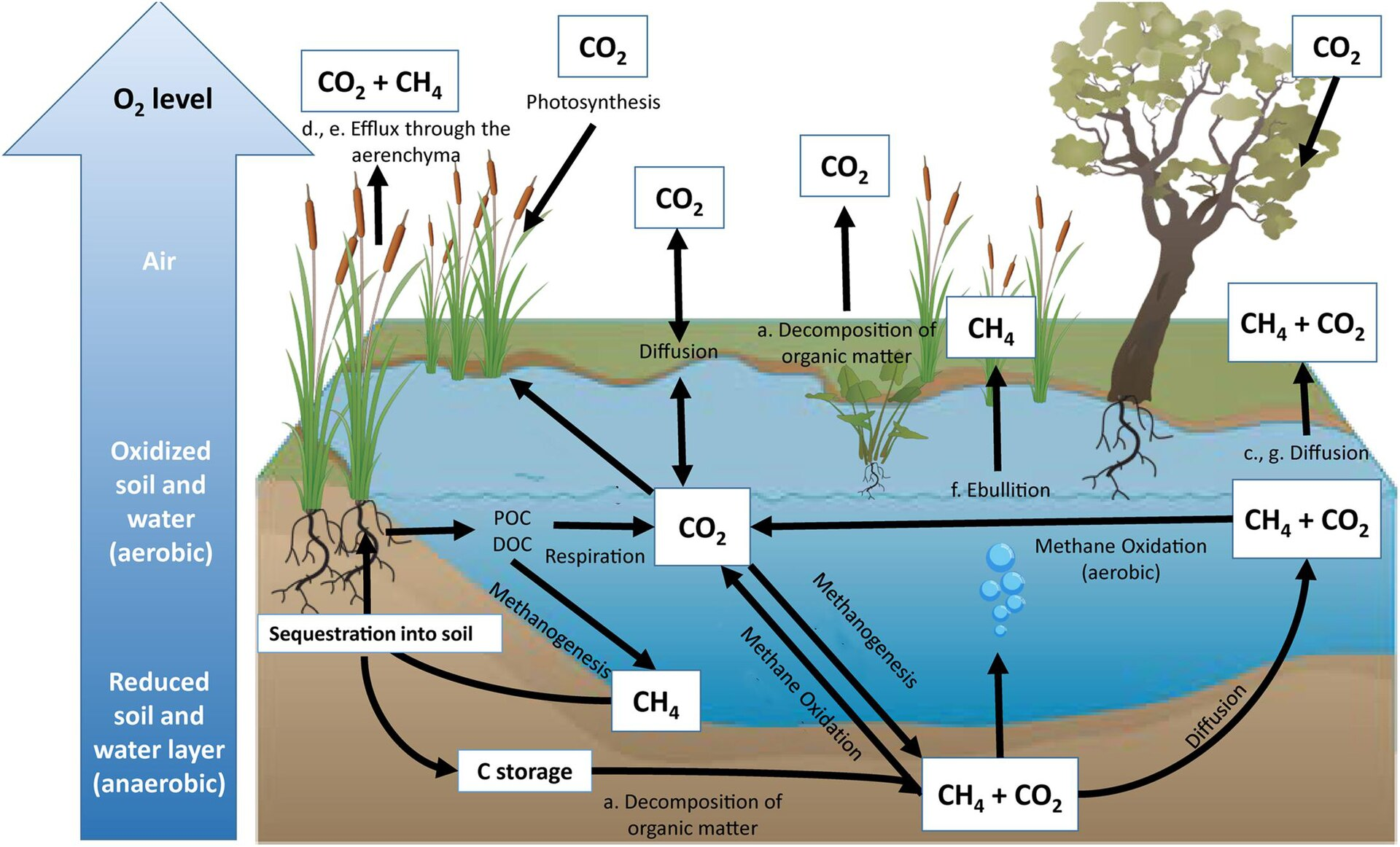
This figure shows how ecosystems such as wetlands can act as both carbon sinks and carbon sources. It highlights exchanges among vegetation, soil, water, and atmospheric gases. It is especially helpful for understanding why land-use change can shift a system from storage to release. Source
Oceans and ocean acidification
· The oceans absorb CO₂ by dissolving it.
· Oceans therefore act as a major carbon sink, but human emissions are occurring faster than oceans can absorb them.
· More dissolved CO₂ lowers ocean pH and causes ocean acidification.
· Ocean acidification harms marine organisms that need calcium carbonate for shells and skeletons.
· Key examples: molluscs and reef-building corals.
· Common exam chain: more atmospheric CO₂ → more dissolved CO₂ → lower pH → reduced calcification → weaker shells/coral skeletons.
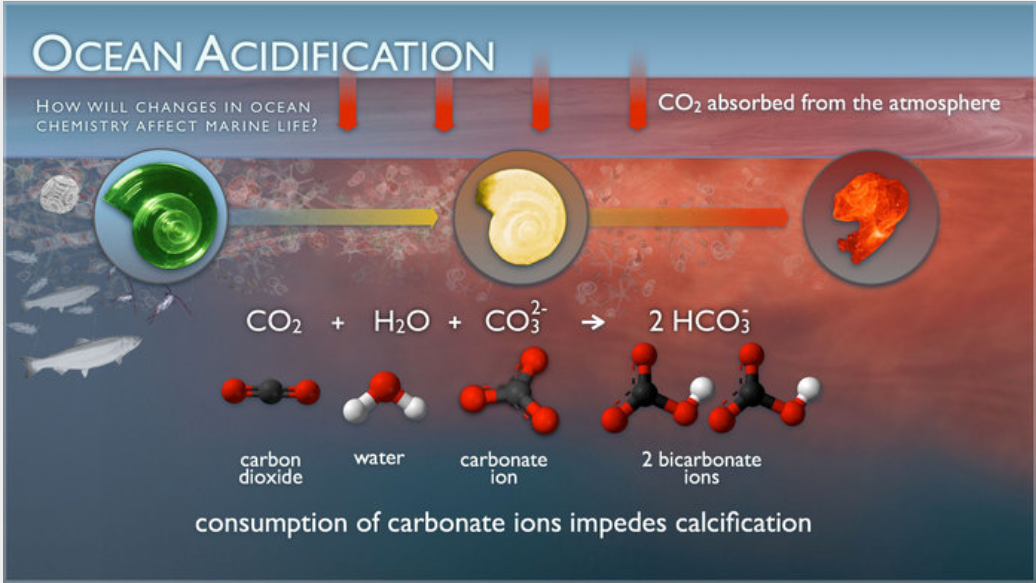
This NOAA illustration shows how rising CO₂ changes ocean chemistry and makes shell and skeleton formation harder for marine organisms. It is ideal for linking the carbon cycle to a direct ecological impact. Revise it as a clear cause-and-effect sequence. Source
Reducing human impacts on the carbon cycle
· ESS expects named measures to reduce disruption of the carbon cycle.
· Examples: low-carbon technologies, reduced fossil-fuel burning, reduced deforestation, less soil disruption, reforestation, artificial carbon sequestration.
· Good evaluation point: prevention is usually more sustainable than relying only on technological fixes.
· In 6-mark/9-mark responses, connect each measure to a specific part of the cycle it changes.
Nitrogen cycle: stores and processes
· The nitrogen cycle has both organic and inorganic stores.
· Organic nitrogen stores = proteins and other nitrogen-containing compounds in living organisms and dead organic matter.
· Inorganic nitrogen stores = atmospheric nitrogen, ammonia, nitrites, nitrates in soil and water.
· The atmosphere contains a huge store of dinitrogen (), but most plants cannot use it directly.
· Bacteria are essential to the nitrogen cycle.
· Key named processes: nitrogen fixation, nitrification, denitrification, decomposition, ammonification.
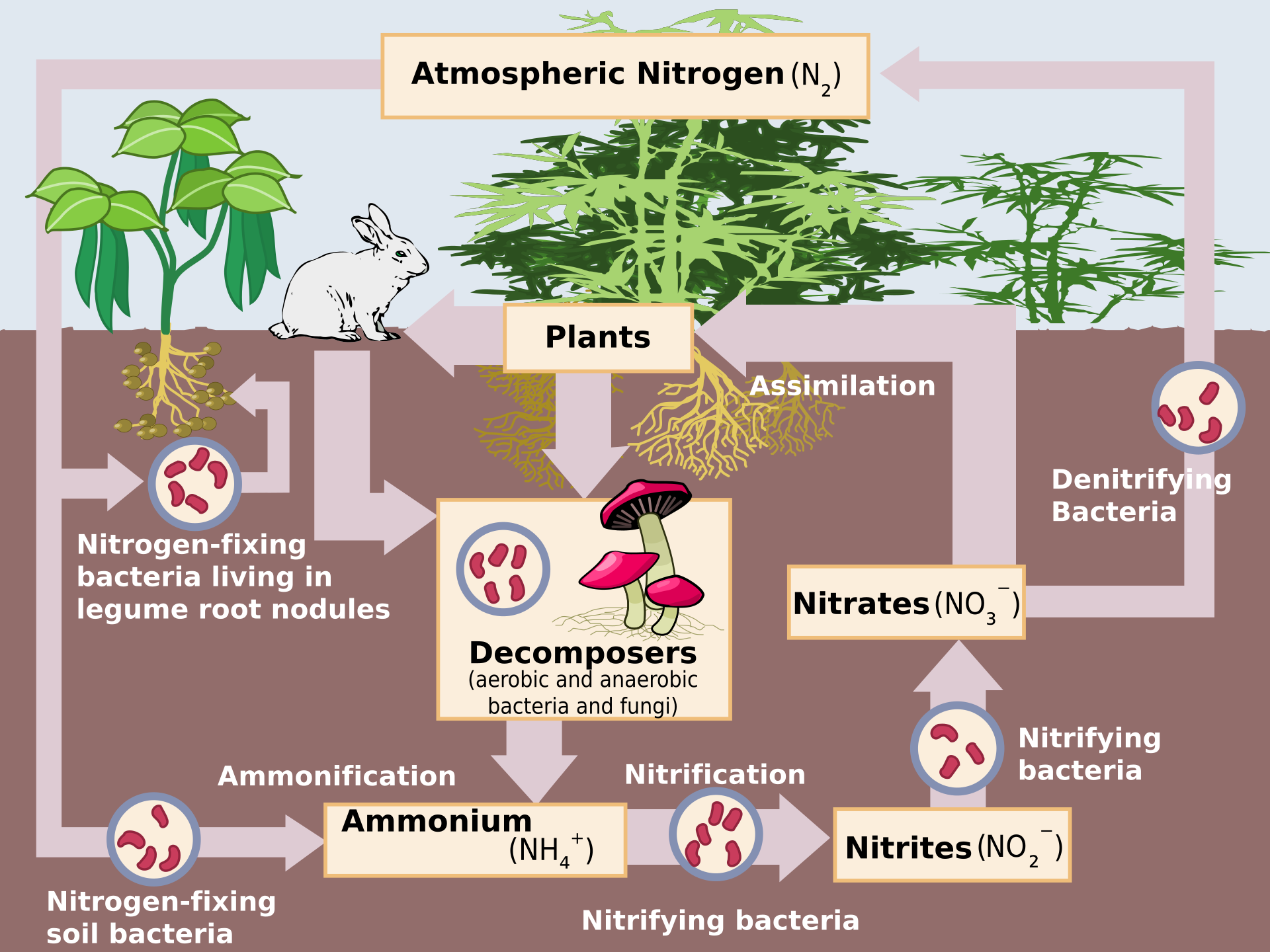
This diagram maps the main nitrogen stores and the bacterial processes linking them, including fixation, nitrification, and denitrification. It is especially useful for distinguishing organic from inorganic nitrogen stores. Practise tracing one nitrogen atom through the full cycle. Source
Essential bacterial roles in the nitrogen cycle
· Nitrogen fixation = conversion of atmospheric into ammonia.
· Nitrification = conversion of ammonia/ammonium into nitrites and then nitrates.
· Denitrification = conversion of nitrates back to atmospheric nitrogen.
· Decomposition breaks down organic nitrogen in dead matter.
· Ammonification converts nitrogen in organic matter/amino acids into ammonium.
· Plants mainly absorb nitrogen as mineral ions from soil, especially nitrates.
· Exam tip: do not confuse nitrogen fixation with mineral uptake by plants.
Conditions affecting the nitrogen cycle
· Denitrification only occurs in anaerobic conditions, such as waterlogged soils.
· Waterlogged soils can reduce plant growth because useful nitrogen is lost and leaching may increase.
· Some plants form mutualistic relationships with nitrogen-fixing bacteria.
· These plants gain a competitive advantage where nitrogen is a limiting factor.
· Typical examples include legumes with root nodules containing nitrogen-fixing bacteria.
Nitrogen flows and human disruption
· Key flows include mineral uptake by producers, consumption, excretion, death, decomposition, and ammonification.
· Human activities that alter the cycle: deforestation, agriculture, aquaculture, urbanization.
· Main human impact = adding large amounts of reactive nitrogen through fertilizers and intensive land use.
· This can increase nitrate concentrations in ecosystems and destabilize Earth systems.
· Nitrogen disruption is important because the planetary boundary for the nitrogen cycle has been crossed.
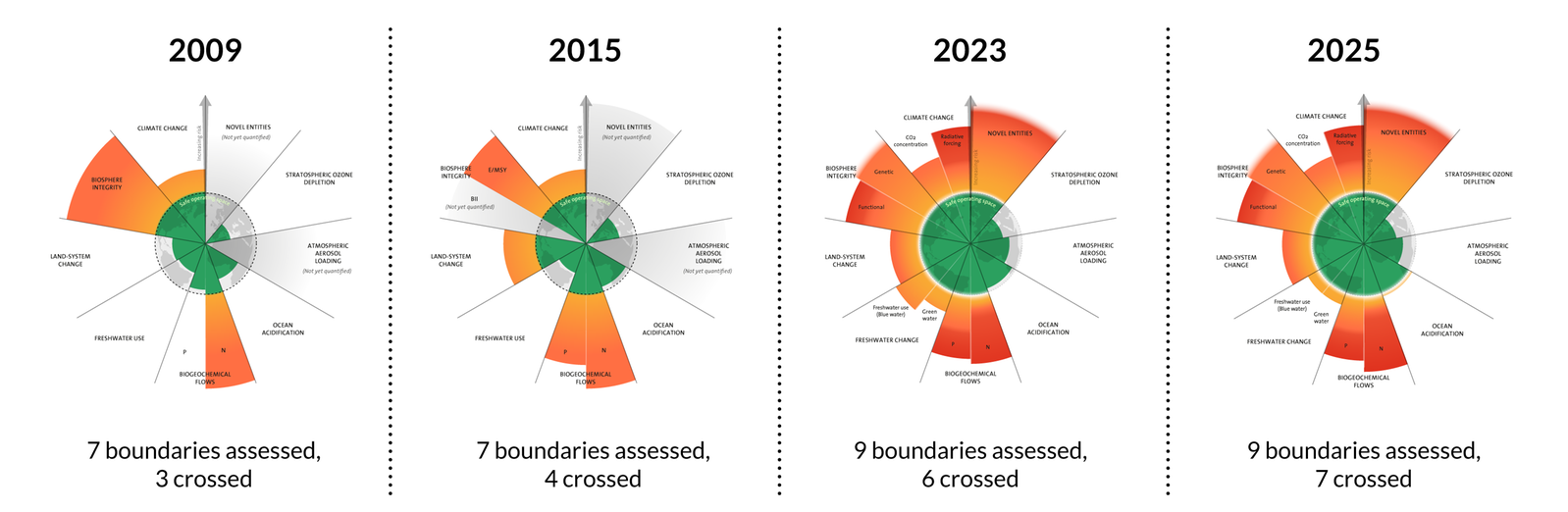
This graphic places biogeochemical flows alongside the other planetary boundaries and shows why excess nitrogen is a global systems issue, not just a farm-scale problem. It helps you connect local fertilizer use to planetary-scale risk. Use it when revising evaluation points about sustainability. Source
Haber process and fertilizer
· The Haber process industrially produces ammonia from nitrogen and hydrogen for fertilizer manufacture.
· Main advantage = much higher crop yield and improved food production.
· Main disadvantages = energy demand, dependence on industrial inputs, and contribution to nitrogen-cycle disruption.
· Strong exam point: the Haber process improves short-term food security but can reduce long-term sustainability if fertilizer use is uncontrolled.
· This is a classic advantages vs disadvantages evaluation question.
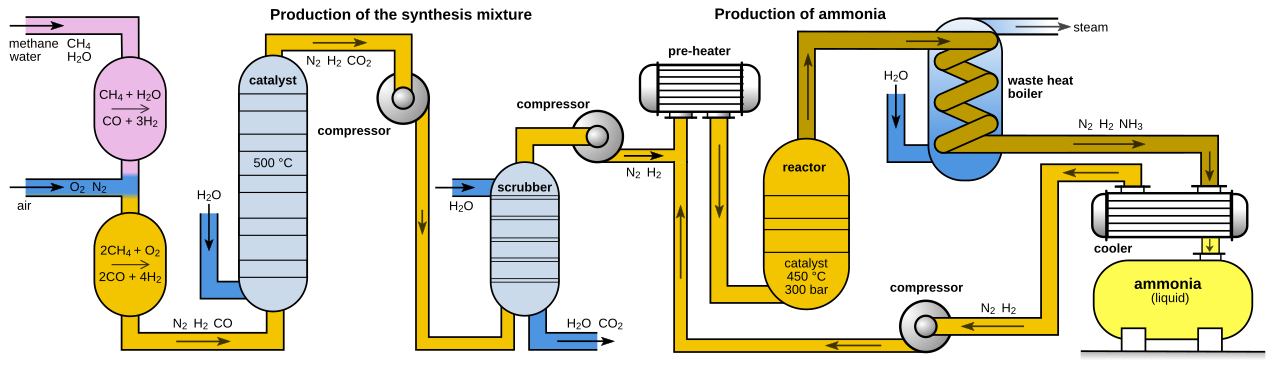
This process diagram shows how the Haber-Bosch process converts nitrogen and hydrogen into ammonia for fertilizer. It is useful for linking industrial chemistry to changes in the nitrogen cycle. Revise it together with the trade-off between higher yield and lower sustainability. Source
Checklist: can you do this?
· Distinguish between store, sink, source, transfer, and transformation.
· Draw and annotate a carbon cycle and nitrogen cycle systems diagram.
· Explain how ecosystems can act as carbon sinks, stores, or sources using named examples.
· Interpret how human activities such as burning fossil fuels, deforestation, and fertilizer use disrupt cycles.
· Evaluate at least one management strategy for the carbon cycle and one for the nitrogen cycle.
HL only: deeper carbon cycle content
· The lithosphere contains major carbon stores in fossil fuels and carbonate rocks such as limestone.
· Carbon in these stores can have residence times of hundreds of millions of years.
· Reef-building corals and molluscs contain calcium carbonate in hard parts that may later become fossilized in limestone.
· Limestone is the largest carbon store in Earth systems.
· In past geological eras, partially decomposed plants became fossilized into coal.
· Partially decomposed marine organisms became fossilized into oil and natural gas in porous rocks.
· Significant fossil-fuel stores took tens of millions of years to accumulate.
· Methane is produced from dead organic matter in anaerobic conditions by methanogenic bacteria.
· Examples of methanogenic environments: swamps, rice paddies, ruminant stomachs.
· Methane has an atmospheric residence time of about 10 years and is later oxidized to CO₂.
· Methane is a potent greenhouse gas, so HL answers should link it to climate change as well as the carbon cycle.
HL only: deeper nitrogen cycle content
· Human-driven increases in nitrates have pushed the nitrogen biogeochemical cycle beyond a planetary boundary.
· This raises the risk of irreversible changes to Earth systems.
· The major driver identified in the syllabus is global dependence on inorganic fertilizers for crop production.
· HL responses should emphasize that solving this requires global collaboration, not just local action.
· Useful solution ideas: more efficient fertilizer use, reducing uncontrolled industrial/agricultural nitrogen release, and shifting toward more sustainable nutrient management.
Exam-ready synthesis
· The best ESS answers connect process, human cause, ecological effect, and management response.
· Carbon-cycle question structure: name store/flow → explain disruption → explain impact → propose measure.
· Nitrogen-cycle question structure: name bacterial process → explain human addition of reactive nitrogen → explain sustainability problem → evaluate solution.
· Repeated big idea: human activity speeds up flows that were naturally slow, overwhelming the ability of systems to remain in equilibrium.
· Final takeaway: biogeochemical cycles are essential for ecosystem stability, and their disruption is a major sustainability challenge.

Shubhi is a seasoned educational specialist with a sharp focus on IB, A-level, GCSE, AP, and MCAT sciences. With 6+ years of expertise, she excels in advanced curriculum guidance and creating precise educational resources, ensuring expert instruction and deep student comprehension of complex science concepts.
Shubhi is a seasoned educational specialist with a sharp focus on IB, A-level, GCSE, AP, and MCAT sciences. With 6+ years of expertise, she excels in advanced curriculum guidance and creating precise educational resources, ensuring expert instruction and deep student comprehension of complex science concepts.