Core idea
Water moves by osmosis across a partially permeable membrane from a less concentrated solution to a more concentrated solution.
Always describe direction in terms of solute concentration or water potential, not “water concentration”.
Key comparison terms:
Hypotonic = surrounding solution has lower solute concentration than the cell.
Hypertonic = surrounding solution has higher solute concentration than the cell.
Isotonic = surrounding solution has equal solute concentration to the cell.
In an isotonic solution, water still moves both ways, but there is dynamic equilibrium and no net movement.
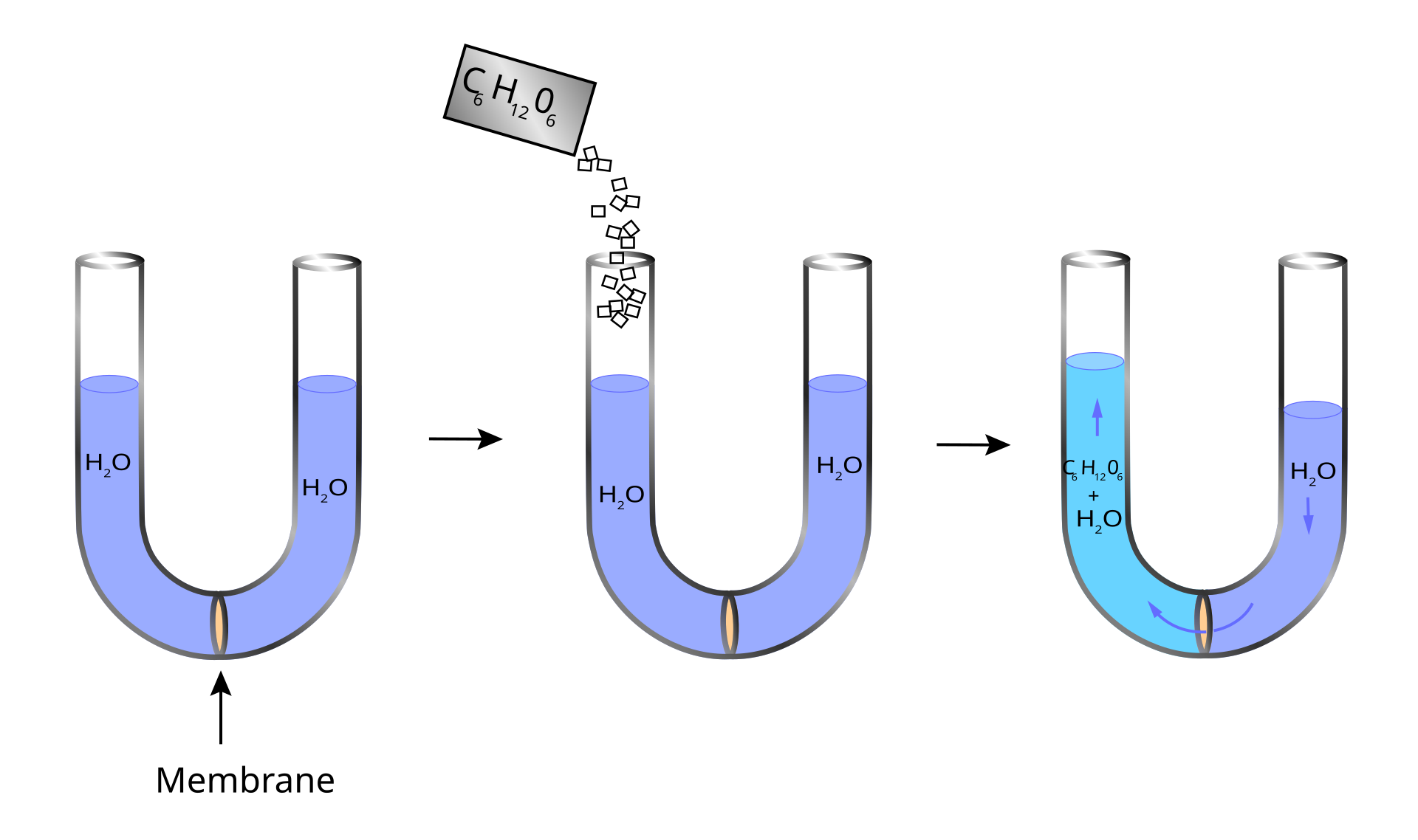
This diagram shows osmosis across a partially permeable membrane in a U-tube. It is useful for linking solute addition to net water movement and the development of osmotic pressure. Source
Solvation and why solutes affect water movement
Water is a polar solvent.
Solvation happens when water molecules surround solute particles.
Water forms hydrogen bonds with some solutes.
Water is also attracted to positive ions and negative ions because of its polarity.
When water interacts with solutes, some of its potential energy becomes less available, helping explain why adding solute lowers water potential.
Exam link: solutes change the direction of osmosis because they change the water potential gradient.
Osmosis into and out of cells
Osmosis = net movement of water through a partially permeable membrane.
Water moves:
from hypotonic → hypertonic conditions
from higher water potential → lower water potential
If a cell is placed in a hypotonic environment, water tends to enter the cell.
If a cell is placed in a hypertonic environment, water tends to leave the cell.
If surroundings are isotonic, there is no net change in cell size or mass.
Common exam trap: the membrane is partially permeable to water, but not equally permeable to solutes.
Effects on cells without a cell wall
In a hypotonic medium, water enters animal cells, causing swelling and possibly bursting (lysis).
In a hypertonic medium, water leaves animal cells, causing shrinkage.
In red blood cells, shrinkage in a hypertonic solution is called crenation.
Cells without a wall are more vulnerable because they lack a rigid outer layer to resist expansion.
Freshwater unicellular organisms use contractile vacuoles to remove excess water that enters by osmosis.
In multicellular organisms, tissue fluids are normally kept isotonic to prevent harmful water gain or loss by cells.
Effects on cells with a cell wall
In a hypotonic medium, water enters plant cells.
The vacuole expands and the contents press against the wall, producing turgor pressure.
A turgid plant cell helps support non-woody plant tissues.
In a hypertonic medium, water leaves the plant cell.
The plasma membrane pulls away from the cell wall: this is plasmolysis.
Plant tissues lose firmness when cells are no longer fully turgid.
Big contrast:
Animal cells may burst in hypotonic solutions.
Plant cells become turgid instead because the cell wall resists expansion.
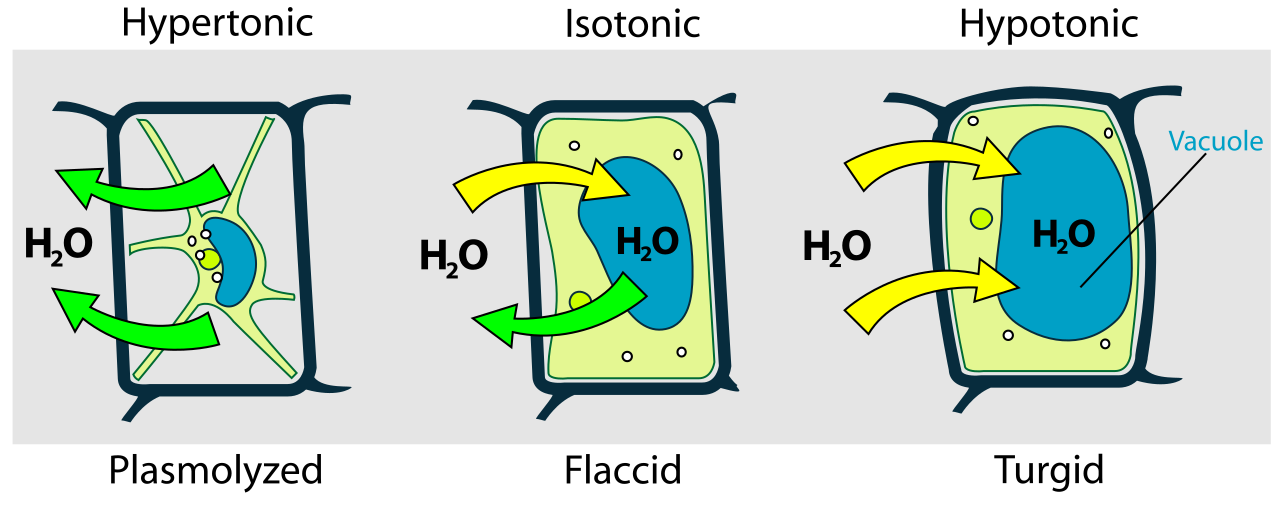
This image compares plant cells in different osmotic conditions, showing turgid, flaccid, and plasmolysed states. It is especially useful for linking water movement to turgor pressure and support in plant tissues. Source
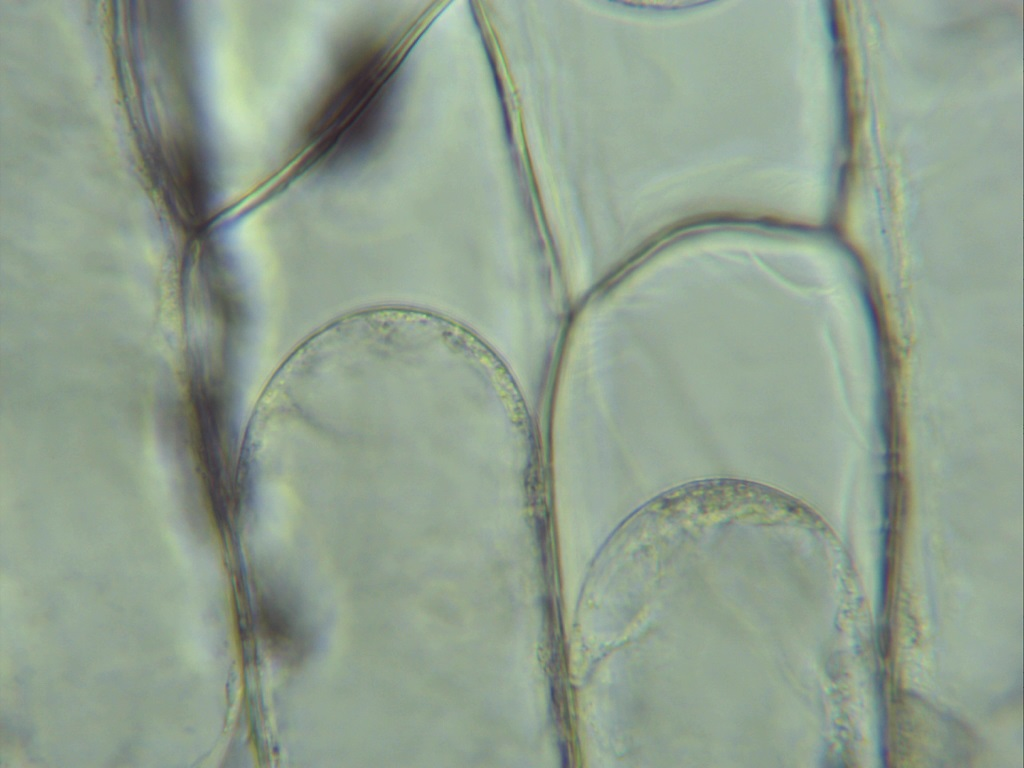
This micrograph shows a real plant cell after water has left the cell in a hypertonic solution. It helps you recognise plasmolysis in practical work and image-based exam questions. Source
Practical: plant tissue in different solute concentrations
You may be asked to use plant tissue such as potato cylinders or other plant samples.
Measure initial and final mass or initial and final length after soaking tissue in solutions of different concentration.
Calculate change in mass or percentage change in mass.
Plot results against solute concentration.
The isotonic concentration is where there is no net change in mass/length.
This isotonic point estimates the tissue’s internal solute concentration.
Repeats improve reliability; use standard deviation or standard error where given.
Standard error bars can help judge how reliable comparisons are.
Exam skill: identify where a graph crosses 0% change and use that as the isotonic point.
Medical applications of isotonic solutions
Isotonic solutions are used when cells must not gain or lose water overall.
Example: intravenous fluids such as 0.9% saline.
These help restore fluid volume without causing major osmotic shifts into or out of cells.
Another example: bathing organs for transplantation in an isotonic solution.
Reason: prevents cells from swelling, shrinking, or being damaged before transplant.
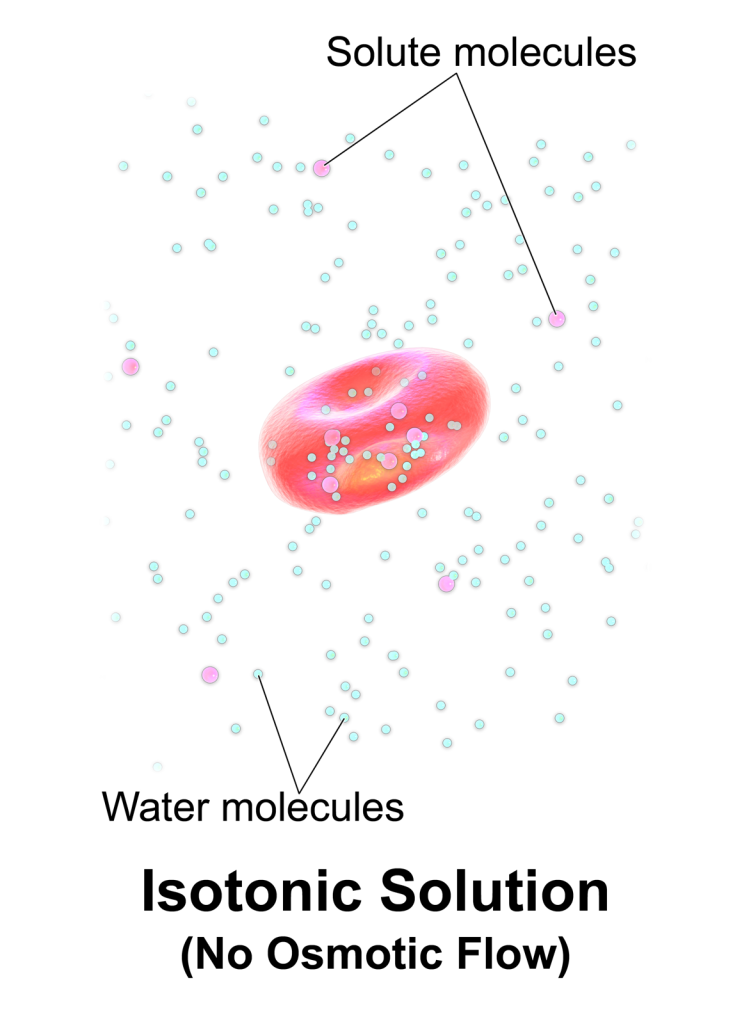
These figures show why isotonic IV fluids do not cause net osmotic movement into or out of cells, while hypotonic and hypertonic fluids do. This is a strong applied example for medical uses of isotonic solutions. Source
HL only: Water potential
Water potential (Ψ) = potential energy of water per unit volume.
Water potential is measured relative to pure water at atmospheric pressure and 20°C.
Pure water has the highest water potential, defined as 0.
The units are usually kilopascals (kPa).
Water moves from higher water potential to lower water potential.
Adding solute makes water potential more negative.
Pressure can raise water potential.
In plant cells with walls:
Ψw = Ψs + Ψp
Ψw = water potential
Ψs = solute potential
Ψp = pressure potential
Solute potential (Ψs) is zero or negative.
Pressure potential (Ψp) is usually positive in turgid plant cells.
In xylem, pressure potential can be negative because water is under tension.
In a hypotonic solution, water enters plant tissue, Ψp rises, and cells become turgid.
In a hypertonic solution, water leaves plant tissue, Ψp falls, and cells may become plasmolysed.
For HL explanations, always link water movement to both solute potential and pressure potential, not just concentration words.
High-yield comparisons
Hypotonic outside cell:
animal cell → swells / may burst
plant cell → turgid
Hypertonic outside cell:
animal cell → shrinks / crenates
plant cell → plasmolysed
Isotonic outside cell:
animal cell → normal size
plant cell → no net movement of water, often flaccid rather than turgid
More solute = lower (more negative) water potential
Water moves down a water potential gradient
Common exam traps
Do not say water moves from high water concentration to low water concentration if the syllabus expects wording in terms of solute concentration or water potential.
Isotonic does not mean no movement; it means no net movement.
Hypertonic / hypotonic are relative terms and must be compared to the cell or another solution.
Plasmolysis only applies to cells with cell walls.
Crenation is used for animal cells / red blood cells, not plant cells.
For HL, remember pressure potential can oppose the effect of solute potential.
Checklist: can you do this?
Predict the direction of net water movement for cells in hypotonic, hypertonic, and isotonic solutions.
Explain the difference between swelling/lysis, crenation, turgor, and plasmolysis.
Interpret practical data to find the isotonic concentration from changes in mass or length.
Use standard deviation / standard error and error bars when judging reliability of osmosis data.
HL: Apply Ψw = Ψs + Ψp to explain water movement in plant cells and xylem.
Ultra-compact memory lines
Hypotonic = water enters.
Hypertonic = water leaves.
Isotonic = dynamic equilibrium.
Plant cell + water in = turgid.
Plant cell + water out = plasmolysed.
Animal cell + water out = crenated.
Water moves from higher Ψ to lower Ψ.

Shubhi is a seasoned educational specialist with a sharp focus on IB, A-level, GCSE, AP, and MCAT sciences. With 6+ years of expertise, she excels in advanced curriculum guidance and creating precise educational resources, ensuring expert instruction and deep student comprehension of complex science concepts.
Shubhi is a seasoned educational specialist with a sharp focus on IB, A-level, GCSE, AP, and MCAT sciences. With 6+ years of expertise, she excels in advanced curriculum guidance and creating precise educational resources, ensuring expert instruction and deep student comprehension of complex science concepts.